Phosphorus Starvation- and Zinc Excess-Induced Astragalus sinicus AsZIP2 Zinc Transporter Is Suppressed by Arbuscular Mycorrhizal Symbiosis
- PMID: 34829181
- PMCID: PMC8623892
- DOI: 10.3390/jof7110892
Phosphorus Starvation- and Zinc Excess-Induced Astragalus sinicus AsZIP2 Zinc Transporter Is Suppressed by Arbuscular Mycorrhizal Symbiosis
Abstract
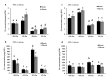
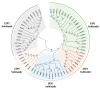
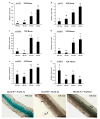
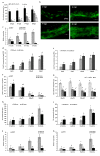
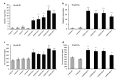
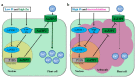
Zinc (Zn) is one of the most essential micronutrients for plant growth and metabolism, but Zn excess can impair many basic metabolic processes in plant cells. In agriculture, crops often experience low phosphate (Pi) and high Zn double nutrient stresses because of inordinate agro-industrial activities, while the dual benefit of arbuscular mycorrhizal (AM) fungi protects plants from experiencing both deficient and toxic nutrient stresses. Although crosstalk between Pi and Zn nutrients in plants have been extensively studied at the physiological level, the molecular basis of how Pi starvation triggers Zn over-accumulation in plants and how AM plants coordinately modulate the Pi and Zn nutrient homeostasis remains to be elucidated. Here, we report that a novel AsZIP2 gene, a Chinese milk vetch (Astragalus sinicus) member of the ZIP gene family, participates in the interaction between Pi and Zn nutrient homeostasis in plants. Phylogenetic analysis revealed that this AsZIP2 protein was closely related to the orthologous Medicago MtZIP2 and Arabidopsis AtZIP2 transporters. Gene expression analysis indicated that AsZIP2 was highly induced in roots by Pi starvation or Zn excess yet attenuated by arbuscular mycorrhization in a Pi-dependent manner. Subcellular localization and heterologous expression experiments further showed that AsZIP2 encoded a functional plasma membrane-localized transporter that mediated Zn uptake in yeast. Moreover, overexpression of AsZIP2 in A. sinicus resulted in the over-accumulation of Zn concentration in roots at low Pi or excessive Zn concentrations, whereas AsZIP2 silencing lines displayed an even more reduced Zn concentration than control lines under such conditions. Our results reveal that the AsZIP2 transporter functioned in Zn over-accumulation in roots during Pi starvation or high Zn supply but was repressed by AM symbiosis in a Pi-dependent manner. These findings also provide new insights into the AsZIP2 gene acting in the regulation of Zn homeostasis in mycorrhizal plants through Pi signal.
Keywords: AsZIP2 transporter; Astragalus sinicus; ZIP gene family; Zinc; arbuscular mycorrhizal fungi; crosstalk between Pi-Zn; phosphate.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




Similar articles
-
A plasma membrane zinc transporter from Medicago truncatula is up-regulated in roots by Zn fertilization, yet down-regulated by arbuscular mycorrhizal colonization.Plant Mol Biol. 2003 Jul;52(5):1077-88. doi: 10.1023/a:1025479701246. Plant Mol Biol. 2003. PMID: 14558666
-
Functional analysis of the novel mycorrhiza-specific phosphate transporter AsPT1 and PHT1 family from Astragalus sinicus during the arbuscular mycorrhizal symbiosis.New Phytol. 2013 May;198(3):836-852. doi: 10.1111/nph.12188. Epub 2013 Feb 27. New Phytol. 2013. PMID: 23442117
-
The auxin-inducible phosphate transporter AsPT5 mediates phosphate transport and is indispensable for arbuscule formation in Chinese milk vetch at moderately high phosphate supply.Environ Microbiol. 2020 Jun;22(6):2053-2079. doi: 10.1111/1462-2920.14952. Epub 2020 Mar 6. Environ Microbiol. 2020. PMID: 32079042
-
Interactions Between Phosphorus, Zinc, and Iron Homeostasis in Nonmycorrhizal and Mycorrhizal Plants.Front Plant Sci. 2019 Sep 26;10:1172. doi: 10.3389/fpls.2019.01172. eCollection 2019. Front Plant Sci. 2019. PMID: 31616454 Free PMC article. Review.
-
Phytohormones, miRNAs, and peptide signals integrate plant phosphorus status with arbuscular mycorrhizal symbiosis.Curr Opin Plant Biol. 2019 Aug;50:132-139. doi: 10.1016/j.pbi.2019.05.004. Epub 2019 Jun 16. Curr Opin Plant Biol. 2019. PMID: 31212139 Review.
Cited by
-
Arbuscular mycorrhiza mitigates zinc stress on Eucalyptus grandis through regulating metal tolerance protein gene expression and ionome uptake.Front Plant Sci. 2022 Nov 7;13:1022696. doi: 10.3389/fpls.2022.1022696. eCollection 2022. Front Plant Sci. 2022. PMID: 36420037 Free PMC article.
References
-
- Marschner H. Marschner’s Mineral Nutrition of Higher Plants. Academic Press; Cambridge, MA, USA: 1995. - DOI
-
- Marichali A., Dallali S., Ouerghemmi S., Sebei H., Hosni K. Germination, morpho-physiological and biochemical responses of coriander (Coriandrum sativum L.) to zinc excess. Ind. Crop. Prod. 2014;55:248–257. doi: 10.1016/j.indcrop.2014.02.033. - DOI
Grants and funding
- NZ2021025/Laboratory of Lingnan Modern Agriculture Project
- 221144/High-level Talent Start Funding Project of South China Agricultural University
- 201904020022/Major Project of Science and Technology of Guangzhou
- 31800092, 32071639/National Natural Science Foundation of China
- 2018A030313141/Natural Science Foundation of Guang-dong Province in China
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

