Preliminary Results of an Ongoing Prospective Clinical Trial on the Use of 68Ga-PSMA and 68Ga-DOTA-RM2 PET/MRI in Staging of High-Risk Prostate Cancer Patients
- PMID: 34829417
- PMCID: PMC8622332
- DOI: 10.3390/diagnostics11112068
Preliminary Results of an Ongoing Prospective Clinical Trial on the Use of 68Ga-PSMA and 68Ga-DOTA-RM2 PET/MRI in Staging of High-Risk Prostate Cancer Patients
Abstract
The aim of the present study is to investigate the synergic role of 68Ga-PSMA PET/MRI and 68Ga-DOTA-RM2 PET/MRI in prostate cancer (PCa) staging. We present pilot data on twenty-two patients with biopsy-proven PCa that underwent 68Ga-PSMA PET/MRI for staging purposes, with 19/22 also undergoing 68Gaa-DOTA-RM2 PET/MRI. TNM classification based on image findings was performed and quantitative imaging parameters were collected for each scan. Furthermore, twelve patients underwent radical prostatectomy with the availability of histological data that were used as the gold standard to validate intraprostatic findings. A DICE score between regions of interest manually segmented on the primary tumour on 68Ga-PSMA PET, 68Ga-DOTA-RM2 PET and on T2 MRI was computed. All imaging modalities detected the primary PCa in 18/19 patients, with 68Ga-DOTA-RM2 PET not detecting any lesion in 1/19 patients. In the remaining patients, 68Ga-PSMA and MRI were concordant. Seven patients presented seminal vesicles involvement on MRI, with two of these being also detected by 68Ga-PSMA, and 68Ga-DOTA-RM2 PET being negative. Regarding extraprostatic disease, 68Ga-PSMA PET, 68Ga-DOTA-RM2 PET and MRI resulted positive in seven, four and five patients at lymph-nodal level, respectively, and at a bone level in three, zero and one patients, respectively. These preliminary results suggest the potential complementary role of 68Ga-PSMA PET, 68Ga-DOTA-RM2 PET and MRI in PCa characterization during the staging phase.
Keywords: PET/MRI; PSMA; RM2; hybrid imaging; multimodal imaging; prostate cancer.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
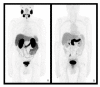

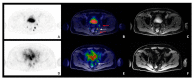
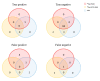
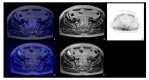
References
-
- Kaufmann S., Kruck S., Gatidis S., Hepp T., Thaiss W.M., Hennenlotter J., Schwenck J., Scharpf M., Nikolaou K., Stenzl A., et al. Simultaneous whole-body PET/MRI with integrated multiparametric MRI for primary staging of high-risk prostate cancer. World J. Urol. 2020;38:2513–2521. doi: 10.1007/s00345-019-03066-1. - DOI - PubMed
-
- Mottet N., van den Bergh R.C., Briers E., van den Broeck T., Cumberbatch M.G., De Santis M., Fanti S., Fossati N., Gandaglia G., Gillessen S., et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer—2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur. Urol. 2020;79:243–262. doi: 10.1016/j.eururo.2020.09.042. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

