Serum Organ-Specific Anti-Heart and Anti-Intercalated Disk Autoantibodies as New Autoimmune Markers of Cardiac Involvement in Systemic Sclerosis: Frequency, Clinical and Prognostic Correlates
- PMID: 34829512
- PMCID: PMC8625508
- DOI: 10.3390/diagnostics11112165
Serum Organ-Specific Anti-Heart and Anti-Intercalated Disk Autoantibodies as New Autoimmune Markers of Cardiac Involvement in Systemic Sclerosis: Frequency, Clinical and Prognostic Correlates
Abstract
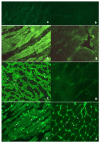
Background: Heart involvement (HInv) in systemic sclerosis (SSc) may relate to myocarditis and is associated with poor prognosis. Serum anti-heart (AHA) and anti-intercalated disk autoantibodies (AIDA) are organ and disease-specific markers of isolated autoimmune myocarditis. We assessed frequencies, clinical correlates, and prognostic impacts of AHA and AIDA in SSc.
Methods: The study included consecutive SSc patients (n = 116, aged 53 ± 13 years, 83.6% females, median disease duration 7 years) with clinically suspected heart involvement (symptoms, abnormal ECG, abnormal troponin I or natriuretic peptides, and abnormal echocardiography). All SSc patients underwent CMR. Serum AHA and AIDA were measured by indirect immunofluorescence in SSc and in control groups of non-inflammatory cardiac disease (NICD) (n = 160), ischemic heart failure (IHF) (n = 141), and normal blood donors (NBD) (n = 270). AHA and AIDA status in SSc was correlated with baseline clinical, diagnostic features, and outcome.
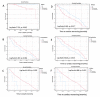
Results: The frequency of AHA was higher in SSc (57/116, 49%, p < 0.00001) than in NICD (2/160, 1%), IHF (2/141, 1%), or NBD (7/270, 2.5%). The frequency of AIDA was higher (65/116, 56%, p < 0.00001) in SSc than in NICD (6/160, 3.75%), IHF (3/141, 2%), or NBD (1/270, 0.37%). AHAs were associated with interstitial lung disease (p = 0.04), history of chest pain (p = 0.026), abnormal troponin (p = 0.006), AIDA (p = 0.000), and current immunosuppression (p = 0.01). AHAs were associated with death (p = 0.02) and overall cardiac events during follow-up (p = 0.017).
Conclusions: The high frequencies of AHA and AIDA suggest a high burden of underdiagnosed autoimmune HInv in SSc. In keeping with the negative prognostic impact of HInv in SSc, AHAs were associated with dismal prognosis.
Keywords: autoantibodies; autoimmunity; myocarditis; prognosis; systemic sclerosis.
Conflict of interest statement
M.M.-C. reports grants and personal fees from Actelion, personal fees from Biogen, personal fees from Bayer, personal fees from Boehringer Ingelheim, personal fees from C.S.L. Behring, and personal fees from Eli-Lilly outside the submitted work. C.B. reports personal fees from Actelion and Eli Lilly, grants from European Scleroderma Trial and Research (EUSTAR) group, New Horizon Fellowship, Foundation for Research in Rheumatology (FOREUM), and Fondazione Italiana per la Ricerca sull’Artrite (FIRA) outside the submitted work. All other authors declare that there are no conflicts of interest.
Figures
References
-
- Bruni C., Buch M.H., Furst D.E., De Luca G., Djokovic A., Dumitru R.B., Giollo A., Polovina M., Steelandt A., Bratis K., et al. Primary systemic sclerosis heart involvement: A systematic literature review and preliminary data-driven, consensus-based WSF/HFA definition. J. Scleroderma Relat. Disord. 2021 doi: 10.1177/23971983211053246. - DOI - PMC - PubMed
-
- Mavrogeni S., Gargani L., Pepe A., Monti L., Markousis-Mavrogenis G., De Santis M., De Marchi D., Koutsogeorgopoulou L., Karabela G., Stavropoulos E., et al. Cardiac magnetic resonance predicts ventricular arrhythmias in scleroderma: The Scleroderma Arrhythmia Clinical Utility Study (SAnCtUS) Rheumatology. 2020;59:1938–1948. - PMC - PubMed
LinkOut - more resources
Full Text Sources

