Aminoguanidine Prevents the Oxidative Stress, Inhibiting Elements of Inflammation, Endothelial Activation, Mesenchymal Markers, and Confers a Renoprotective Effect in Renal Ischemia and Reperfusion Injury
- PMID: 34829595
- PMCID: PMC8614713
- DOI: 10.3390/antiox10111724
Aminoguanidine Prevents the Oxidative Stress, Inhibiting Elements of Inflammation, Endothelial Activation, Mesenchymal Markers, and Confers a Renoprotective Effect in Renal Ischemia and Reperfusion Injury
Abstract
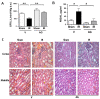
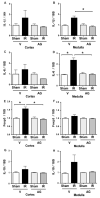
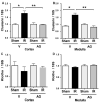
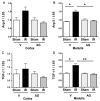
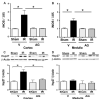
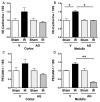
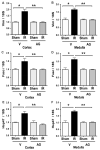
Oxidative stress produces macromolecules dysfunction and cellular damage. Renal ischemia-reperfusion injury (IRI) induces oxidative stress, inflammation, epithelium and endothelium damage, and cessation of renal function. The IRI is an inevitable process during kidney transplantation. Preliminary studies suggest that aminoguanidine (AG) is an antioxidant compound. In this study, we investigated the antioxidant effects of AG (50 mg/kg, intraperitoneal) and its association with molecular pathways activated by IRI (30 min/48 h) in the kidney. The antioxidant effect of AG was studied measuring GSSH/GSSG ratio, GST activity, lipoperoxidation, iNOS, and Hsp27 levels. In addition, we examined the effect of AG on elements associated with cell survival, inflammation, endothelium, and mesenchymal transition during IRI. AG prevented lipid peroxidation, increased GSH levels, and recovered the GST activity impaired by IRI. AG was associated with inhibition of iNOS, Hsp27, endothelial activation (VE-cadherin, PECAM), mesenchymal markers (vimentin, fascin, and HSP47), and inflammation (IL-1β, IL-6, Foxp3, and IL-10) upregulation. In addition, AG reduced kidney injury (NGAL, clusterin, Arg-2, and TFG-β1) and improved kidney function (glomerular filtration rate) during IRI. In conclusion, we found new evidence of the antioxidant properties of AG as a renoprotective compound during IRI. Therefore, AG is a promising compound to treat the deleterious effect of renal IRI.
Keywords: aminoguanidine; antioxidants; ischemia-reperfusion injury; oxidative stress; renal protection.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

