Thermodynamic Studies of Interactions between Sertraline Hydrochloride and Randomly Methylated β-Cyclodextrin Molecules Supported by Circular Dichroism Spectroscopy and Molecular Docking Results
- PMID: 34830239
- PMCID: PMC8620473
- DOI: 10.3390/ijms222212357
Thermodynamic Studies of Interactions between Sertraline Hydrochloride and Randomly Methylated β-Cyclodextrin Molecules Supported by Circular Dichroism Spectroscopy and Molecular Docking Results
Abstract
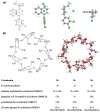
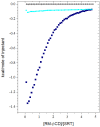
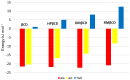
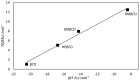
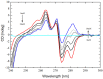
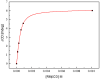
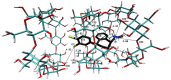
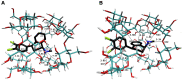
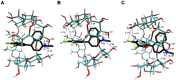
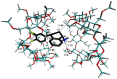
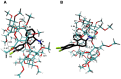
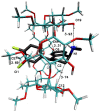
The interaction between sertraline hydrochloride (SRT) and randomly methylated β-cyclodextrin (RMβCD) molecules have been investigated at 298.15 K under atmospheric pressure. The method used-Isothermal Titration Calorimetry (ITC) enabled to determine values of the thermodynamic functions like the enthalpy (ΔH), the entropy (ΔS) and the Gibbs free energy (ΔG) of binding for the examined system. Moreover, the stoichiometry coefficient of binding (n) and binding/association constant (K) value have been calculated from the experimental results. The obtained outcome was compared with the data from the literature for other non-ionic βCD derivatives interacting with SRT and the enthalpy-entropy compensation were observed and interpreted. Furthermore, the connection of RMβCD with SRT was characterized by circular dichroism spectroscopy (CD) and complexes of βCD derivatives with SRT were characterized through the computational studies with the use of molecular docking (MD).
Keywords: circular dichroism; isothermal titration calorimetry; methylated β-cyclodextrin; molecular docking; sertraline hydrochloride; β-cyclodextrin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Dalla Bella M., Szejtli J. Cyclodextrins. Drugs Future. 1983;8:391–394.
-
- Szente L., Puskás I., Sohajda T., Varga E., Vass P., Nagy Z.K., Farkas A., Várnai B., Béni S., Hazai E. Sulfobutylether-beta-cyclodextrin-enabled antiviral remdesivir: Characterization of electrospun- and lyophilized formulations. Carbohydr. Polym. 2021;264:118011. doi: 10.1016/j.carbpol.2021.118011. - DOI - PMC - PubMed
-
- Sursyakova V.V., Levdansky V.A., Rubaylo A.I. Thermodynamic parameters for the complexation of water-soluble betulin derivatives with (2-hydroxypropyl)-β-cyclodextrin determined by affinity capillary electrophoresis. J. Mol. Liq. 2019;283:325–331. doi: 10.1016/j.molliq.2019.03.092. - DOI
-
- Lin Z.-Y., Liu Y.-X., Kou S.-B., Wang B.-L., Shi J.-H. Characterization of the inclusion interaction of ethinyloestradiol with β-cyclodextrin and hydroxypropyl-β-cyclodextrin: Multi-spectroscopic and molecular modeling methods. J. Mol. Liq. 2020;311:113290. doi: 10.1016/j.molliq.2020.113290. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

