Aesculetin Accelerates Osteoblast Differentiation and Matrix-Vesicle-Mediated Mineralization
- PMID: 34830274
- PMCID: PMC8621655
- DOI: 10.3390/ijms222212391
Aesculetin Accelerates Osteoblast Differentiation and Matrix-Vesicle-Mediated Mineralization
Abstract
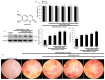
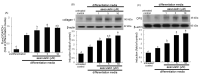
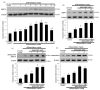
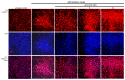
The imbalance between bone resorption and bone formation in favor of resorption results in bone loss and deterioration of bone architecture. Osteoblast differentiation is a sequential event accompanying biogenesis of matrix vesicles and mineralization of collagen matrix with hydroxyapatite crystals. Considerable efforts have been made in developing naturally-occurring plant compounds, preventing bone pathologies, or enhancing bone regeneration. Coumarin aesculetin inhibits osteoporosis through hampering the ruffled border formation of mature osteoclasts. However, little is known regarding the effects of aesculetin on the impairment of matrix vesicle biogenesis. MC3T3-E1 cells were cultured in differentiation media with 1-10 μM aesculetin for up to 21 days. Aesculetin boosted the bone morphogenetic protein-2 expression, and alkaline phosphatase activation of differentiating MC3T3-E1 cells. The presence of aesculetin strengthened the expression of collagen type 1 and osteoprotegerin and transcription of Runt-related transcription factor 2 in differentiating osteoblasts for 9 days. When ≥1-5 μM aesculetin was added to differentiating cells for 15-18 days, the induction of non-collagenous proteins of bone sialoprotein II, osteopontin, osteocalcin, and osteonectin was markedly enhanced, facilitating the formation of hydroxyapatite crystals and mineralized collagen matrix. The induction of annexin V and PHOSPHO 1 was further augmented in ≥5 μM aesculetin-treated differentiating osteoblasts for 21 days. In addition, the levels of tissue-nonspecific alkaline phosphatase and collagen type 1 were further enhanced within the extracellular space and on matrix vesicles of mature osteoblasts treated with aesculetin, indicating matrix vesicle-mediated bone mineralization. Finally, aesculetin markedly accelerated the production of thrombospondin-1 and tenascin C in mature osteoblasts, leading to their adhesion to preformed collagen matrix. Therefore, aesculetin enhanced osteoblast differentiation, and matrix vesicle biogenesis and mineralization. These findings suggest that aesculetin may be a potential osteo-inductive agent preventing bone pathologies or enhancing bone regeneration.
Keywords: aesculetin; collagen mineralization; hydroxyapatite; matrix vesicles; non-collagenous proteins; osteoblast differentiation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
The dual-effects of LaCl₃ on the proliferation, osteogenic differentiation, and mineralization of MC3T3-E1 cells.Biol Trace Elem Res. 2012 Dec;150(1-3):433-40. doi: 10.1007/s12011-012-9486-6. Epub 2012 Aug 14. Biol Trace Elem Res. 2012. PMID: 22886987
-
JNK activity is essential for Atf4 expression and late-stage osteoblast differentiation.J Bone Miner Res. 2009 Mar;24(3):398-410. doi: 10.1359/jbmr.081107. J Bone Miner Res. 2009. PMID: 19016586
-
Inductive effects of dexamethasone on the gene expression of Cbfa1, Osterix and bone matrix proteins during differentiation of cultured primary rat osteoblasts.J Mol Histol. 2004 Jan;35(1):3-10. doi: 10.1023/b:hijo.0000020883.33256.fe. J Mol Histol. 2004. PMID: 15323344
-
Collagen and non-collagenous proteins molecular crosstalk in the pathophysiology of osteoporosis.Cytokine Growth Factor Rev. 2019 Oct;49:59-69. doi: 10.1016/j.cytogfr.2019.09.001. Epub 2019 Sep 13. Cytokine Growth Factor Rev. 2019. PMID: 31543432 Review.
-
Matrix Vesicle-Mediated Mineralization and Osteocytic Regulation of Bone Mineralization.Int J Mol Sci. 2022 Sep 1;23(17):9941. doi: 10.3390/ijms23179941. Int J Mol Sci. 2022. PMID: 36077336 Free PMC article. Review.
Cited by
-
Dietary Collagen Hydrolysates Retard Estrogen Deficiency-Induced Bone Loss through Blocking Osteoclastic Activation and Enhancing Osteoblastic Matrix Mineralization.Biomedicines. 2022 Jun 10;10(6):1382. doi: 10.3390/biomedicines10061382. Biomedicines. 2022. PMID: 35740404 Free PMC article.
-
The Role of Thrombospondins in Osteoarthritis: from Molecular Mechanisms to Therapeutic Potential.Int J Biol Sci. 2025 Mar 3;21(5):2346-2359. doi: 10.7150/ijbs.103343. eCollection 2025. Int J Biol Sci. 2025. PMID: 40083685 Free PMC article. Review.
-
Metabolomics Analysis of Phenolic Composition and Content in Five Pear Cultivars Leaves.Plants (Basel). 2024 Sep 7;13(17):2513. doi: 10.3390/plants13172513. Plants (Basel). 2024. PMID: 39273997 Free PMC article.
-
Estrogen Receptor 1 (ESR1) and the Wnt/β-Catenin Pathway Mediate the Effect of the Coumarin Derivative Umbelliferon on Bone Mineralization.Nutrients. 2022 Aug 5;14(15):3209. doi: 10.3390/nu14153209. Nutrients. 2022. PMID: 35956385 Free PMC article.
-
Aesculetin Suppresses Airway Epithelial Barrier Disruption Induced by Inhalation of Urban Coarse Particulate: Involvement of Neutrophil Elastase and Protease-Activated Receptor‑2.ACS Omega. 2025 May 15;10(20):20866-20874. doi: 10.1021/acsomega.5c02198. eCollection 2025 May 27. ACS Omega. 2025. PMID: 40454068 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

