Cutaneous Adverse Reactions Associated with SARS-CoV-2 Vaccines
- PMID: 34830627
- PMCID: PMC8618511
- DOI: 10.3390/jcm10225344
Cutaneous Adverse Reactions Associated with SARS-CoV-2 Vaccines
Abstract
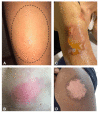
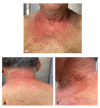
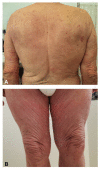
Many patients are receiving SARS-CoV-2 vaccinations, which have been associated with a variety of adverse effects. Cutaneous adverse reactions to SARS-CoV-2 vaccinations have been progressively reported, but they have not been reviewed according to their morphological clinical patterns. The objective of this review was to summarize the existing data concerning the cutaneous adverse reactions following SARS-CoV-2 vaccines and group them according to common morphological and pathogenetic patterns. We reviewed the English language literature up to 15 August 2021, using predefined keywords to identify the relevant studies evaluating cutaneous adverse reactions associated with SARS-CoV-2 vaccines. We search for recurrent morphological patterns sharing clinical signs and symptoms and physio-pathological mechanisms. Timing to onset following the first or booster dose of the vaccine, predisposing conditions, therapeutic management, and outcome were also collected. Among the dermatological manifestations associated with SARS-CoV-2 vaccinations, we distinguished: (1) new onset reactions and (2) flares of preexisting dermatoses. The most common were injection site reactions, affecting 30-70% and generally mild or moderate. Small case series or single case reports included filler reactions, exanthemas, vascular lesions, urticaria, eczematous dermatitis, autoimmune bullous reactions, and severe cutaneous adverse reactions. In addition, the exacerbation of chronic immuno-mediated dermatoses (mainly psoriasis and atopic dermatitis) and reactivations of herpes infection were reported. The cutaneous reactions were generally mild, self-limiting, and resembled common cutaneous drug eruptions and/or COVID-19 skin manifestations.
Keywords: COVID-19; cutaneous adverse reaction; exanthema; safety; vaccines.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

