Recent Advancement in Chitosan-Based Nanoparticles for Improved Oral Bioavailability and Bioactivity of Phytochemicals: Challenges and Perspectives
- PMID: 34833334
- PMCID: PMC8617804
- DOI: 10.3390/polym13224036
Recent Advancement in Chitosan-Based Nanoparticles for Improved Oral Bioavailability and Bioactivity of Phytochemicals: Challenges and Perspectives
Abstract
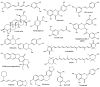
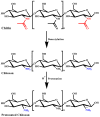
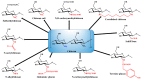
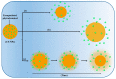
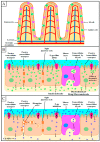
The excellent therapeutic potential of a variety of phytochemicals in different diseases has been proven by extensive studies throughout history. However, most phytochemicals are characterized by a high molecular weight, poor aqueous solubility, limited gastrointestinal permeability, extensive pre-systemic metabolism, and poor stability in the harsh gastrointestinal milieu. Therefore, loading of these phytochemicals in biodegradable and biocompatible nanoparticles (NPs) might be an effective approach to improve their bioactivity. Different nanocarrier systems have been developed in recent decades to deliver phytochemicals. Among them, NPs based on chitosan (CS) (CS-NPs), a mucoadhesive, non-toxic, and biodegradable polysaccharide, are considered the best nanoplatform for the oral delivery of phytochemicals. This review highlights the oral delivery of natural products, i.e., phytochemicals, encapsulated in NPs prepared from a natural polymer, i.e., CS, for improved bioavailability and bioactivity. The unique properties of CS for oral delivery such as its mucoadhesiveness, non-toxicity, excellent stability in the harsh environment of the GIT, good solubility in slightly acidic and alkaline conditions, and ability to enhance intestinal permeability are discussed first, and then the outcomes of various phytochemical-loaded CS-NPs after oral administration are discussed in detail. Furthermore, different challenges associated with the oral delivery of phytochemicals with CS-NPs and future directions are also discussed.
Keywords: bioactivity; bioavailability; chitosan nanoparticles; mucoadhesion; phytochemicals.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Fatima M., Iqubal M.K., Iqubal A., Kaur H., Gilani S.J., Rahman M.H., Ahmadi A., Rizwanullah M. Current insight into the therapeutic potential of phytocompounds and their nanoparticle-based systems for effective management of lung cancer. Anticancer Agents Med. Chem. 2021;22:1–19. doi: 10.2174/1871520621666210708123750. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

