Precision Medicine in Systemic Mastocytosis
- PMID: 34833353
- PMCID: PMC8623914
- DOI: 10.3390/medicina57111135
Precision Medicine in Systemic Mastocytosis
Abstract
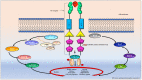
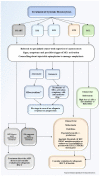
Mastocytosis is a rare hematological neoplasm characterized by the proliferation of abnormal clonal mast cells (MCs) in different cutaneous and extracutaneous organs. Its diagnosis is based on well-defined major and minor criteria, including the pathognomonic dense infiltrate of MCs detected in bone marrow (BM), elevated serum tryptase level, abnormal MCs CD25 expression, and the identification of KIT D816V mutation. The World Health Organization (WHO) classification subdivides mastocytosis into a cutaneous form (CM) and five systemic variants (SM), namely indolent/smoldering (ISM/SSM) and advanced SM (AdvSM) including aggressive SM (ASM), SM associated to hematological neoplasms (SM-AHN), and mast cell leukemia (MCL). More than 80% of patients with SM carry a somatic point mutation of KIT at codon 816, which may be targeted by kinase inhibitors. The presence of additional somatic mutations detected by next generation sequencing analysis may impact prognosis and drive treatment strategy, which ranges from symptomatic drugs in indolent forms to kinase-inhibitors active on KIT. Allogeneic stem cell transplant (SCT) may be considered in selected SM cases. Here, we review the clinical, diagnostic, and therapeutic issues of SM, with special emphasis on the translational implications of SM genetics for a precision medicine approach in clinical practice.
Keywords: KIT; genetics; precision medicine; systemic mastocytosis; tyrosine kinase inhibitor.
Conflict of interest statement
G.G. declares personal consulting fees from Janssen, Abbvie Astra-Zeneca, Beigene, and Incyte. A.P. declares personal consulting fees from Ariad, Sanofi, and Takeda.
Figures


References
-
- Arber D.A., Orazi A., Hasserjian R., Thiele J., Borowitz M.J., Beau M.M., Bloomfield C.D., Cazzola M., Vardiman J.M. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leuke-mia. Blood. 2016;127:2391–2405. doi: 10.1182/blood-2016-03-643544. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

