The Roles of Amphibian (Xenopus laevis) Macrophages during Chronic Frog Virus 3 Infections
- PMID: 34835105
- PMCID: PMC8621048
- DOI: 10.3390/v13112299
The Roles of Amphibian (Xenopus laevis) Macrophages during Chronic Frog Virus 3 Infections
Abstract
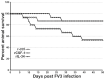
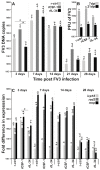
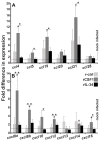
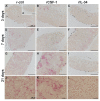
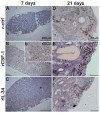
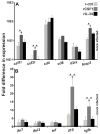
Infections by Frog Virus 3 (FV3) and other ranavirus genus members are significantly contributing to global amphibian decline. The Xenopus laevis frog is an ideal research platform upon which to study the roles of distinct frog leukocyte populations during FV3 infections. Frog macrophages (MΦs) are integrally involved during FV3 infection, as they facilitate viral dissemination and persistence but also participate in immune defense against this pathogen. In turn, MΦ differentiation and functionality depend on the colony-stimulating factor-1 receptor (CSF-1R), which is ligated by CSF-1 and iterleukin-34 (IL-34) cytokines. Our past work indicated that X. laevis CSF-1 and IL-34 give rise to morphologically and functionally distinct frog MΦ subsets, and that these CSF-1- and IL-34-MΦs respectively confer susceptibility and antiviral resistance to FV3. Because FV3 targets the frog kidneys and establishes chronic infections therein, presently we examined the roles of the frog CSF-1- and IL-34-MΦs in seeding and maintaining these chronic kidney infections. Our findings indicate that the frog CSF-1-MΦs result in more prominent kidney FV3 infections, which develop into greater reservoirs of lingering FV3 marked by infiltrating leukocytes, fibrosis, and overall immunosuppressive states. Moreover, the antiviral effects of IL-34-MΦs are short-lived and are lost as FV3 infections progress.
Keywords: amphibian; interferons; intestine; myeloid cells; ranavirus.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Differentiation-dependent antiviral capacities of amphibian (Xenopus laevis) macrophages.J Biol Chem. 2018 Feb 2;293(5):1736-1744. doi: 10.1074/jbc.M117.794065. Epub 2017 Dec 19. J Biol Chem. 2018. PMID: 29259133 Free PMC article.
-
Prominent amphibian (Xenopus laevis) tadpole type III interferon response to the frog virus 3 ranavirus.J Virol. 2015 May;89(9):5072-82. doi: 10.1128/JVI.00051-15. Epub 2015 Feb 25. J Virol. 2015. PMID: 25717104 Free PMC article.
-
The amphibian (Xenopus laevis) colony-stimulating factor-1 and interleukin-34-derived macrophages possess disparate pathogen recognition capacities.Dev Comp Immunol. 2019 Sep;98:89-97. doi: 10.1016/j.dci.2019.04.011. Epub 2019 Apr 25. Dev Comp Immunol. 2019. PMID: 31029710
-
Immune evasion strategies of ranaviruses and innate immune responses to these emerging pathogens.Viruses. 2012 Jul;4(7):1075-92. doi: 10.3390/v4071075. Epub 2012 Jun 28. Viruses. 2012. PMID: 22852041 Free PMC article. Review.
-
Antiviral immunity in amphibians.Viruses. 2011 Nov;3(11):2065-2086. doi: 10.3390/v3112065. Epub 2011 Oct 31. Viruses. 2011. PMID: 22163335 Free PMC article. Review.
Cited by
-
Xenopus laevis (Daudin, 1802) as a Model Organism for Bioscience: A Historic Review and Perspective.Biology (Basel). 2023 Jun 20;12(6):890. doi: 10.3390/biology12060890. Biology (Basel). 2023. PMID: 37372174 Free PMC article. Review.
-
The amphibian immune system.Philos Trans R Soc Lond B Biol Sci. 2023 Jul 31;378(1882):20220123. doi: 10.1098/rstb.2022.0123. Epub 2023 Jun 12. Philos Trans R Soc Lond B Biol Sci. 2023. PMID: 37305914 Free PMC article. Review.
-
Primary regulatory T cell activator FOXP3 is present across Amphibia.Immunogenetics. 2025 Feb 13;77(1):15. doi: 10.1007/s00251-025-01372-0. Immunogenetics. 2025. PMID: 39945843
-
Detection of Frog Virus 3 by Integrating RPA-CRISPR/Cas12a-SPM with Deep Learning.ACS Omega. 2023 Aug 25;8(36):32555-32564. doi: 10.1021/acsomega.3c02929. eCollection 2023 Sep 12. ACS Omega. 2023. PMID: 37720737 Free PMC article.
-
Recombinant SpTransformer proteins are functionally diverse for binding and phagocytosis by three subtypes of sea urchin phagocytes.Front Immunol. 2024 Apr 29;15:1372904. doi: 10.3389/fimmu.2024.1372904. eCollection 2024. Front Immunol. 2024. PMID: 38742116 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

