Alphavirus-Driven Interferon Gamma (IFNg) Expression Inhibits Tumor Growth in Orthotopic 4T1 Breast Cancer Model
- PMID: 34835178
- PMCID: PMC8620866
- DOI: 10.3390/vaccines9111247
Alphavirus-Driven Interferon Gamma (IFNg) Expression Inhibits Tumor Growth in Orthotopic 4T1 Breast Cancer Model
Abstract
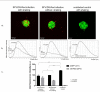
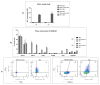
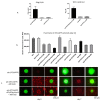
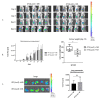
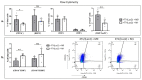
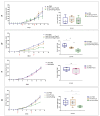
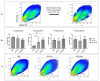
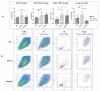
Interferon gamma (IFNg) is a pleiotropic cytokine that can potentially reprogram the tumor microenvironment; however, the antitumor immunomodulatory properties of IFNg still need to be validated due to variable therapeutic outcomes in preclinical and clinical studies. We developed a replication-deficient Semliki Forest virus vector expressing IFNg (SFV/IFNg) and evaluated its immunomodulatory antitumor potential in vitro in a model of 3D spheroids and in vivo in an immunocompetent 4T1 mouse breast cancer model. We demonstrated that SFV-derived, IFN-g-stimulated bone marrow macrophages can be used to acquire the tumoricidal M1 phenotype in 3D nonattached conditions. Coculturing SFV/IFNg-infected 4T1 spheroids with BMDMs inhibited spheroid growth. In the orthotopic 4T1 mouse model, intratumoral administration of SFV/IFNg virus particles alone or in combination with the Pam3CSK4 TLR2/1 ligand led to significant inhibition of tumor growth compared to the administration of the control SFV/Luc virus particles. Analysis of the composition of intratumoral lymphoid cells isolated from tumors after SFV/IFNg treatment revealed increased CD4+ and CD8+ and decreased T-reg (CD4+/CD25+/FoxP3+) cell populations. Furthermore, a significant decrease in the populations of cells bearing myeloid cell markers CD11b, CD38, and CD206 was observed. In conclusion, the SFV/IFNg vector induces a therapeutic antitumor T-cell response and inhibits myeloid cell infiltration in treated tumors.
Keywords: CD38; Pam3CSK4; alphavirus; bone-marrow-derived macrophages; cancer immunotherapy; interferon gamma; spheroids; viral vectors.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Neoadjuvant administration of Semliki Forest virus expressing interleukin-12 combined with attenuated Salmonella eradicates breast cancer metastasis and achieves long-term survival in immunocompetent mice.BMC Cancer. 2015 Sep 7;15:620. doi: 10.1186/s12885-015-1618-x. BMC Cancer. 2015. PMID: 26347489 Free PMC article.
-
Generation and Functional In Vitro Analysis of Semliki Forest Virus Vectors Encoding TNF-α and IFN-γ.Front Immunol. 2017 Nov 30;8:1667. doi: 10.3389/fimmu.2017.01667. eCollection 2017. Front Immunol. 2017. PMID: 29276511 Free PMC article.
-
Short-Term Local Expression of a PD-L1 Blocking Antibody from a Self-Replicating RNA Vector Induces Potent Antitumor Responses.Mol Ther. 2019 Nov 6;27(11):1892-1905. doi: 10.1016/j.ymthe.2019.09.016. Epub 2019 Sep 16. Mol Ther. 2019. PMID: 31563534 Free PMC article.
-
Alphavirus vectors for gene therapy applications.Curr Gene Ther. 2001 May;1(1):19-29. doi: 10.2174/1566523013349039. Curr Gene Ther. 2001. PMID: 12109136 Review.
-
Alphaviruses and their derived vectors as anti-tumor agents.Curr Cancer Drug Targets. 2004 Nov;4(7):597-607. doi: 10.2174/1568009043332754. Curr Cancer Drug Targets. 2004. PMID: 15578917 Review.
Cited by
-
Perspective Technologies of Vaccination: Do We Still Need Old Vaccines?Vaccines (Basel). 2022 Jun 2;10(6):891. doi: 10.3390/vaccines10060891. Vaccines (Basel). 2022. PMID: 35746498 Free PMC article.
-
Fibronectin Functions as a Selective Agonist for Distinct Toll-like Receptors in Triple-Negative Breast Cancer.Cells. 2022 Jun 30;11(13):2074. doi: 10.3390/cells11132074. Cells. 2022. PMID: 35805158 Free PMC article.
-
An overview of "Chronic viral infection and cancer, openings for vaccines" virtual symposium of the TechVac Network - December 16-17, 2021.Infect Agent Cancer. 2022 Jul 8;17(Suppl 2):28. doi: 10.1186/s13027-022-00436-0. Infect Agent Cancer. 2022. PMID: 35804391 Free PMC article.
-
Cancer Spheroids and Organoids as Novel Tools for Research and Therapy: State of the Art and Challenges to Guide Precision Medicine.Cells. 2023 Mar 24;12(7):1001. doi: 10.3390/cells12071001. Cells. 2023. PMID: 37048073 Free PMC article. Review.
-
Proceedings of the Online Conference "Vaccines and Vaccination during and Post COVID Pandemics" (7-9 December 2022).Vaccines (Basel). 2023 Jun 29;11(7):1175. doi: 10.3390/vaccines11071175. Vaccines (Basel). 2023. PMID: 37514990 Free PMC article.
References
-
- Kalanjeri S., Sterman D.H. Gene therapy in interventional pulmonology: Interferon gene delivery with focus on thoracic malignancies. Curr. Respir. Care Rep. 2012;1:54–66. doi: 10.1007/s13665-011-0008-3. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

