A prospective phase-II trial of biweekly docetaxel plus androgen deprivation therapy in patients with previously-untreated metastatic castration-naïve prostate cancer
- PMID: 34839812
- PMCID: PMC8628395
- DOI: 10.1186/s12885-021-09018-6
A prospective phase-II trial of biweekly docetaxel plus androgen deprivation therapy in patients with previously-untreated metastatic castration-naïve prostate cancer
Abstract
Introduction: The aim of this prospective phase II study was to evaluate the efficacy and safety of biweekly docetaxel plus androgen-deprivation therapy (ADT) in patients with metastatic castration-naïve prostate cancer (mCNPC).
Patients and methods: Patients with histologically-proven, previously-untreated mCNPC received ADT plus docetaxel, 40 mg/m2. Docetaxel was repeated every 2 weeks, up to 12 cycles. Endpoints included castration-resistant prostate cancer (CRPC)-free survival, prostate-specific antigen (PSA) response, and safety.
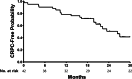
Results: A total of 42 patients were registered and analyzed for final outcomes. Of the 42 patients, 36 (86%) completed the 12 planned cycles of docetaxel plus ADT. During a median follow up of 25 months, all but two patients (95%) achieved a PSA response with a nadir PSA level of 0.42 ng/ml (range 0.01-1280.87). The median CRPC-free survival was 26.4 months (95% confidence interval [CI] 20.9-32.0) with a one-year CRPC-free rate of 79% (33 patients, 95% CI 66-91). Multivariable analysis revealed that the performance status of the Eastern Cooperative Oncology Group 0 was independently associated with longer CRPC-free survival (hazard ratio [HR] 0.27, 95% CI 0.07-0.99). The most common adverse events of any grade were anemia (95%), followed by nail changes (33%), fatigue (29%), and oral mucositis (26%). Severe (grade 3 or higher) adverse events were infrequent: pneumonitis (n = 2), diarrhea (n = 1), and neutropenia (n = 1).
Conclusion: Our results suggest that biweekly docetaxel plus ADT is feasible, and clinical efficacy does not seem to be compromised compared to a standard triweekly docetaxel 75 mg/m2 plus ADT regimen.
© 2021. The Author(s).
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures

Similar articles
-
Effect of Adding Docetaxel to Androgen-Deprivation Therapy in Patients With High-Risk Prostate Cancer With Rising Prostate-Specific Antigen Levels After Primary Local Therapy: A Randomized Clinical Trial.JAMA Oncol. 2019 May 1;5(5):623-632. doi: 10.1001/jamaoncol.2018.6607. JAMA Oncol. 2019. PMID: 30703190 Free PMC article. Clinical Trial.
-
Anticancer Activity and Tolerance of Treatments Received Beyond Progression in Men Treated Upfront with Androgen Deprivation Therapy With or Without Docetaxel for Metastatic Castration-naïve Prostate Cancer in the GETUG-AFU 15 Phase 3 Trial.Eur Urol. 2018 May;73(5):696-703. doi: 10.1016/j.eururo.2017.09.022. Epub 2017 Oct 23. Eur Urol. 2018. PMID: 29074061 Clinical Trial.
-
A retrospective feasibility study of biweekly docetaxel in patients with high-risk metastatic castration-naïve prostate cancer.BMC Urol. 2019 May 3;19(1):30. doi: 10.1186/s12894-019-0463-7. BMC Urol. 2019. PMID: 31053137 Free PMC article.
-
[Treatment situation in metastastic Castration Naive Prostate Cancer (mCRPC) and the implications on clinical routine].Urologe A. 2019 Sep;58(9):1066-1072. doi: 10.1007/s00120-019-0925-2. Urologe A. 2019. PMID: 31041460 Review. German.
-
What kind of patients with castration-naïve prostate cancer can benefit from upfront docetaxel and abiraterone: A systematic review and a network meta-analysis.Urol Oncol. 2018 Dec;36(12):505-517. doi: 10.1016/j.urolonc.2018.09.005. Epub 2018 Oct 11. Urol Oncol. 2018. PMID: 30316578
Cited by
-
Ethnic Pharmacogenomic Differences in the Management of Asian Patients with Metastatic Prostate Cancer.Cancers (Basel). 2022 Jan 14;14(2):407. doi: 10.3390/cancers14020407. Cancers (Basel). 2022. PMID: 35053569 Free PMC article. Review.
-
Effectiveness of Adding Docetaxel to Androgen Deprivation Therapy for Metastatic Hormone-Sensitive Prostate Cancer in Korean Real-World Practice.Yonsei Med J. 2023 Feb;64(2):86-93. doi: 10.3349/ymj.2022.0244. Yonsei Med J. 2023. PMID: 36719015 Free PMC article.
-
Baseline characteristics may impact treatment duration of cabazitaxel in patients with mCRPC: a subanalysis of data from a post-marketing surveillance.Jpn J Clin Oncol. 2024 Jan 7;54(1):97-102. doi: 10.1093/jjco/hyad128. Jpn J Clin Oncol. 2024. PMID: 37807695 Free PMC article.
References
-
- James ND, Sydes MR, Clarke NW, et al. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet. 2016;387:1163–1177. doi: 10.1016/S0140-6736(15)01037-5. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

