Preservation of Eye Movements in Parkinson's Disease Is Stimulus- and Task-Specific
- PMID: 34848498
- PMCID: PMC8802919
- DOI: 10.1523/JNEUROSCI.1690-21.2021
Preservation of Eye Movements in Parkinson's Disease Is Stimulus- and Task-Specific
Abstract
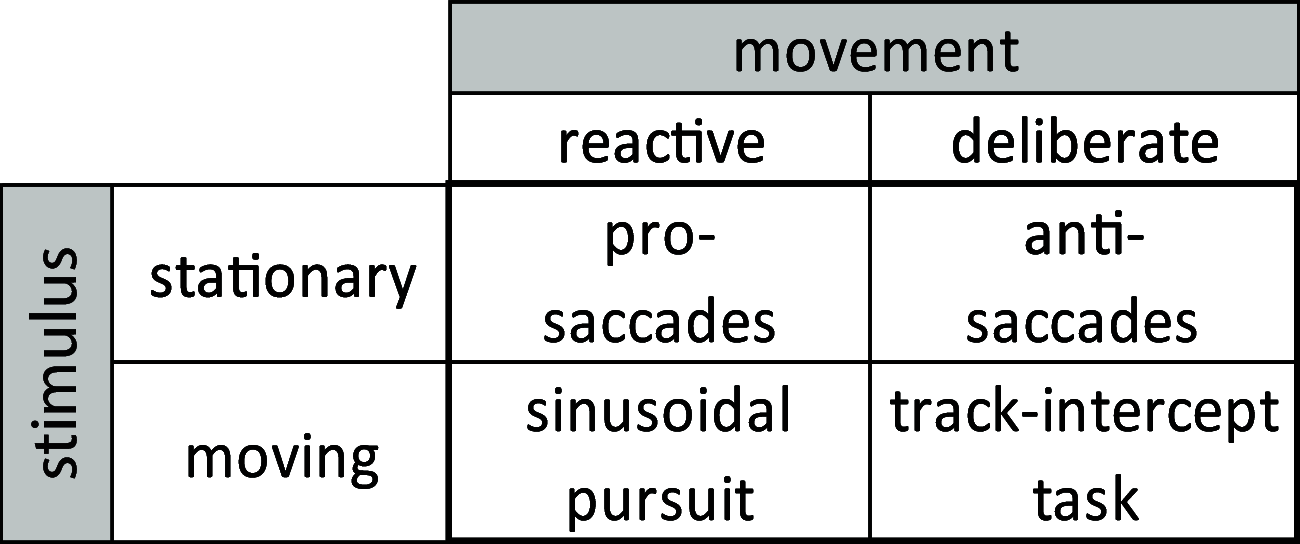
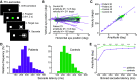
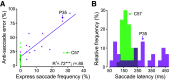
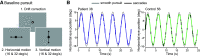
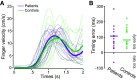
Parkinson's disease (PD) is a neurodegenerative disease that includes motor impairments, such as tremor, bradykinesia, and postural instability. Although eye movement deficits are commonly found in saccade and pursuit tasks, preservation of oculomotor function has also been reported. Here we investigate specific task and stimulus conditions under which oculomotor function in PD is preserved. Sixteen PD patients and 18 healthy, age-matched controls completed a battery of movement tasks that included stationary or moving targets eliciting reactive or deliberate eye movements: pro-saccades, anti-saccades, visually guided pursuit, and rapid go/no-go manual interception. Compared with controls, patients demonstrated systematic impairments in tasks with stationary targets: pro-saccades were hypometric and anti-saccades were incorrectly initiated toward the cued target in ∼35% of trials compared with 14% errors in controls. In patients, task errors were linked to short latency saccades, indicating abnormalities in inhibitory control. However, patients' eye movements in response to dynamic targets were relatively preserved. PD patients were able to track and predict a disappearing moving target and make quick go/no-go decisions as accurately as controls. Patients' interceptive hand movements were slower on average but initiated earlier, indicating adaptive processes to compensate for motor slowing. We conclude that PD patients demonstrate stimulus and task dependency of oculomotor impairments, and we propose that preservation of eye and hand movement function in PD is linked to a separate functional pathway through the superior colliculus-brainstem loop that bypasses the fronto-basal ganglia network. Our results demonstrate that studying oculomotor and hand movement function in PD can support disease diagnosis and further our understanding of disease progression and dynamics.SIGNIFICANCE STATEMENT Eye movements are a promising clinical tool to aid in the diagnosis of movement disorders and to monitor disease progression. Although Parkinson's disease (PD) patients show some oculomotor abnormalities, it is not clear whether previously described eye movement impairments are task-specific. We assessed eye movements in PD under different visual (stationary vs moving targets) and movement (reactive vs deliberate) conditions. We demonstrate that PD patients are able to accurately track moving objects but make inaccurate eye movements toward stationary targets. The preservation of eye movements toward dynamic stimuli might enable patients to accurately act on the predicted motion path of the moving target. These results can inform the development of tools for the rehabilitation or maintenance of functional performance.
Keywords: Parkinson's disease; eye movements; prediction; preservation of function; saccades; smooth pursuit.
Copyright © 2022 the authors.
Figures


Similar articles
-
Deficits in saccadic eye-movement control in Parkinson's disease.Neuropsychologia. 2005;43(5):784-96. doi: 10.1016/j.neuropsychologia.2004.06.026. Neuropsychologia. 2005. PMID: 15721191
-
Subthalamic deep brain stimulation improves smooth pursuit and saccade performance in patients with Parkinson's disease.J Neuroeng Rehabil. 2013 Apr 3;10:33. doi: 10.1186/1743-0003-10-33. J Neuroeng Rehabil. 2013. PMID: 23551890 Free PMC article.
-
The initiation of smooth pursuit eye movements and saccades in normal subjects and in "express-saccade makers".Exp Brain Res. 2002 Jun;144(3):373-84. doi: 10.1007/s00221-002-1059-z. Epub 2002 Apr 13. Exp Brain Res. 2002. PMID: 12021819
-
Clinical application of eye movement tasks as an aid to understanding Parkinson's disease pathophysiology.Exp Brain Res. 2017 May;235(5):1309-1321. doi: 10.1007/s00221-017-4916-5. Epub 2017 Mar 3. Exp Brain Res. 2017. PMID: 28258438 Review.
-
Eye movements in Parkinson's disease: from neurophysiological mechanisms to diagnostic tools.Trends Neurosci. 2024 Jan;47(1):71-83. doi: 10.1016/j.tins.2023.11.001. Epub 2023 Dec 2. Trends Neurosci. 2024. PMID: 38042680 Review.
Cited by
-
Machine Learning and Eye Movements Give Insights into Neurodegenerative Disease Mechanisms.Sensors (Basel). 2023 Feb 14;23(4):2145. doi: 10.3390/s23042145. Sensors (Basel). 2023. PMID: 36850743 Free PMC article. Review.
-
Reflexive eye saccadic parameters in Parkinson's disease.Front Med Technol. 2024 Nov 25;6:1477502. doi: 10.3389/fmedt.2024.1477502. eCollection 2024. Front Med Technol. 2024. PMID: 39654658 Free PMC article.
-
The Role of ΔFosB in the Pathogenesis of Levodopa-Induced Dyskinesia: Mechanisms and Therapeutic Strategies.Mol Neurobiol. 2025 Jun;62(6):7393-7412. doi: 10.1007/s12035-025-04720-z. Epub 2025 Jan 31. Mol Neurobiol. 2025. PMID: 39890697 Review.
-
Digital biomarkers for precision diagnosis and monitoring in Parkinson's disease.NPJ Digit Med. 2024 Aug 21;7(1):218. doi: 10.1038/s41746-024-01217-2. NPJ Digit Med. 2024. PMID: 39169258 Free PMC article. Review.
-
Exploration of sensory-motor tradeoff behavior in Parkinson's disease.Front Hum Neurosci. 2022 Oct 25;16:951313. doi: 10.3389/fnhum.2022.951313. eCollection 2022. Front Hum Neurosci. 2022. PMID: 36393983 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
