A Novel Dibenzoxazepine Attenuates Intracellular Salmonella Typhimurium Oxidative Stress Resistance
- PMID: 34851152
- PMCID: PMC8635125
- DOI: 10.1128/Spectrum.01519-21
A Novel Dibenzoxazepine Attenuates Intracellular Salmonella Typhimurium Oxidative Stress Resistance
Abstract
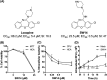
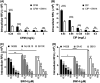
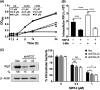
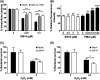
Salmonella enterica serovar Typhimurium is the leading cause of invasive nontyphoidal salmonellosis. Additionally, the emergence of multidrug-resistant S. Typhimurium has further increased the difficulty of controlling its infection. Previously, we showed that an antipsychotic drug, loxapine, suppressed intracellular Salmonella in macrophages. To exploit loxapine's antibacterial activity, we simultaneously evaluated the anti-intracellular Salmonella activity and cytotoxicity of newly synthesized loxapine derivatives using an image-based high-content assay. We identified that SW14 exhibits potent suppressive effects on intramacrophagic S. Typhimurium with an 50% effective concentration (EC50) of 0.5 μM. SW14 also sensitized intracellular Salmonella to ciprofloxacin and cefixime and effectively controlled intracellular multidrug- and fluoroquinolone-resistant S. Typhimurium strains. However, SW14 did not affect bacterial growth in standard microbiological broth or minimal medium that mimics the phagosomal environment. Cellular autophagy blockade by 3-methyladenine (3-MA) or shATG7 elevated the susceptibility of intracellular Salmonella to SW14. Finally, reactive oxygen species (ROS) scavengers reduced the antibacterial efficacy of SW14, but the ROS levels in SW14-treated macrophages were not elevated. SW14 decreased the resistance of outer membrane-compromised S. Typhimurium to H2O2. Collectively, our data indicated that the structure of loxapine can be further optimized to develop new antibacterial agents by targeting bacterial resistance to host oxidative-stress defense. IMPORTANCE The incidence of diseases caused by pathogenic bacteria with resistance to common antibiotics is consistently increasing. In addition, Gram-negative bacteria are particularly difficult to treat with antibiotics, especially those that can invade and proliferate intracellularly. In order to find a new antibacterial compound against intracellular Salmonella, we established a cell-based high-content assay and identified SW14 from the derivatives of the antipsychotic drug loxapine. Our data indicate that SW14 has no effect on free bacteria in the medium but can suppress the intracellular proliferation of multidrug-resistant (MDR) S. Typhimurium in macrophages. We also found that SW14 can suppress the resistance of outer membrane compromised Salmonella to H2O2, and its anti-intracellular Salmonella activity can be reversed by reactive oxygen species (ROS) scavengers. Together, the findings suggest that SW14 might act via a virulence-targeted mechanism and that its structure has the potential to be further developed as a new therapeutic against MDR Salmonella.
Keywords: ROS; antipsychotics; high-content assay; loxapine.
Conflict of interest statement
We declare no conflicts of interest.
Figures
Similar articles
-
Loxapine, an antipsychotic drug, suppresses intracellular multiple-antibiotic-resistant Salmonella enterica serovar Typhimurium in macrophages.J Microbiol Immunol Infect. 2019 Aug;52(4):638-647. doi: 10.1016/j.jmii.2019.05.006. Epub 2019 Jun 8. J Microbiol Immunol Infect. 2019. PMID: 31239204
-
Discovery of antipsychotic loxapine derivatives against intracellular multidrug-resistant bacteria.RSC Med Chem. 2022 Aug 11;13(11):1361-1366. doi: 10.1039/d2md00182a. eCollection 2022 Nov 16. RSC Med Chem. 2022. PMID: 36439974 Free PMC article.
-
Repurposing the tyrosine kinase inhibitor nilotinib for use against intracellular multidrug-resistant Salmonella Typhimurium.J Microbiol Immunol Infect. 2023 Jun;56(3):490-498. doi: 10.1016/j.jmii.2023.01.005. Epub 2023 Jan 17. J Microbiol Immunol Infect. 2023. PMID: 36702646
-
Prevalence and multidrug resistance in Salmonella enterica Typhimurium: an overview in South East Asia.World J Microbiol Biotechnol. 2021 Sep 28;37(11):185. doi: 10.1007/s11274-021-03146-8. World J Microbiol Biotechnol. 2021. PMID: 34580741 Review.
-
Phytochemicals: potential alternative strategy to fight Salmonella enterica serovar Typhimurium.Front Vet Sci. 2023 May 16;10:1188752. doi: 10.3389/fvets.2023.1188752. eCollection 2023. Front Vet Sci. 2023. PMID: 37261108 Free PMC article. Review.
Cited by
-
Virulence potential of Salmonella 1,4, [5],12:i:- strains isolated during decades from different sources in the Southeast region of Brazil.Braz J Microbiol. 2023 Dec;54(4):2827-2843. doi: 10.1007/s42770-023-01145-5. Epub 2023 Oct 10. Braz J Microbiol. 2023. PMID: 37817050 Free PMC article.
-
Discovery of new dibenzodiazepine derivatives as antibacterials against intracellular bacteria.RSC Med Chem. 2023 Dec 11;15(1):283-292. doi: 10.1039/d3md00418j. eCollection 2024 Jan 25. RSC Med Chem. 2023. PMID: 38283231 Free PMC article.
References
-
- Majowicz SE, Musto J, Scallan E, Angulo FJ, Kirk M, O’Brien SJ, Jones TF, Fazil A, Hoekstra RM, Burd ICED, International Collaboration on Enteric Disease “Burden of Illness” Studies. 2010. The global burden of nontyphoidal Salmonella gastroenteritis. Clin Infect Dis 50:882–889. doi:10.1086/650733. - DOI - PubMed
-
- GBD 2017 Non-Typhoidal Salmonella Invasive Disease Collaborators. 2019. The global burden of non-typhoidal Salmonella invasive disease: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Infect Dis 19:1312–1324. 10.1016/S1473-3099(19)30418-9. doi:10.1016/S1473-3099(19)30418-9. - DOI - DOI - PMC - PubMed
-
- Tacconelli E, Carrara E, Savoldi A, Harbarth S, Mendelson M, Monnet DL, Pulcini C, Kahlmeter G, Kluytmans J, Carmeli Y, Ouellette M, Outterson K, Patel J, Cavaleri M, Cox EM, Houchens CR, Grayson ML, Hansen P, Singh N, Theuretzbacher U, Magrini N, Aboderin AO, Al-Abri SS, Awang Jalil N, Benzonana N, Bhattacharya S, Brink AJ, Burkert FR, Cars O, Cornaglia G, Dyar OJ, Friedrich AW, Gales AC, Gandra S, Giske CG, Goff DA, Goossens H, Gottlieb T, Guzman Blanco M, Hryniewicz W, Kattula D, Jinks T, Kanj SS, Kerr L, Kieny M-P, Kim YS, Kozlov RS, Labarca J, Laxminarayan R, Leder K, et al. . 2018. Discovery, research, and development of new antibiotics: the WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect Dis 18:318–327. doi:10.1016/S1473-3099(17)30753-3. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

