Clusterin suppresses invasion and metastasis of testicular seminoma by upregulating COL15a1
- PMID: 34853731
- PMCID: PMC8608570
- DOI: 10.1016/j.omtn.2021.11.004
Clusterin suppresses invasion and metastasis of testicular seminoma by upregulating COL15a1
Abstract
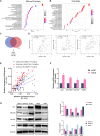
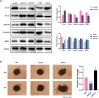
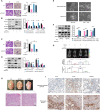
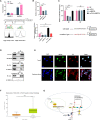
Seminoma is the most common subtype of testicular germ cell tumor, with an increasing incidence worldwide. Clusterin (CLU) expression was found to be downregulated in testicular seminoma in our previous study. We now expanded the sample size, and further indicated that CLU expression correlates with tumor stage. Tcam-2 cell line was used to investigate the CLU function in testicular seminoma, and CLU was found to inhibit the proliferation and metastasis abilities. Besides, extracellular matrix protein COL15a1 was demonstrated as the downstream of CLU to affect the epithelial-mesenchymal transition (EMT) process via competitively binding to DDR1 with COL1A1 and inhibiting the phosphorylation of PYK2. MEF2A was found to interact with CLU and bind to the promoter of COL15a1 and so upregulate its expression. This is the first study using testicular xenografts in situ to simulate testicular seminoma metastatic and proliferative capacities. In conclusion, CLU acts as a tumor suppressor to inhibit the metastasis of testicular seminoma by interacting with MEF2A to upregulate COL15a1 and blocking the EMT process.
Keywords: COL15a1; EMT; MEF2A; clusterin; seminoma; testicular xenografts in situ.
© 2021 The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Testicular orphan receptor 4 promotes tumor progression and implies poor survival through AKT3 regulation in seminoma.Cancer Sci. 2018 Feb;109(2):384-394. doi: 10.1111/cas.13461. Cancer Sci. 2018. PMID: 29197138 Free PMC article.
-
[Correlation between expression of metastasis-related genes and lymph node metastasis in testicular cancer].Hinyokika Kiyo. 2000 Oct;46(10):775-81. Hinyokika Kiyo. 2000. PMID: 11215209 Japanese.
-
Clusterin suppresses spermatogenic cell apoptosis to alleviate diabetes-induced testicular damage by inhibiting autophagy via the PI3K/AKT/mTOR axis.Biol Cell. 2021 Jan;113(1):14-27. doi: 10.1111/boc.202000030. Epub 2020 Nov 4. Biol Cell. 2021. PMID: 32942336
-
The molecular chaperone apolipoprotein J/clusterin as a sensor of oxidative stress: implications in therapeutic approaches - a mini-review.Gerontology. 2013;59(6):514-23. doi: 10.1159/000351207. Epub 2013 May 15. Gerontology. 2013. PMID: 23689375 Review.
-
Metachronous bilateral testicular seminoma developing after an interval of 31 years: case report and review of the literature.Int J Surg Pathol. 2015 Apr;23(2):156-60. doi: 10.1177/1066896914548794. Epub 2014 Aug 29. Int J Surg Pathol. 2015. PMID: 25172883 Review.
Cited by
-
Exploiting potential molecular compounds for treating testicular seminoma by targeting immune related genes.Cell Commun Signal. 2024 Nov 21;22(1):560. doi: 10.1186/s12964-024-01927-w. Cell Commun Signal. 2024. PMID: 39574183 Free PMC article.
-
Emerging RNA-Based Therapeutic and Diagnostic Options: Recent Advances and Future Challenges in Genitourinary Cancers.Int J Mol Sci. 2023 Feb 27;24(5):4601. doi: 10.3390/ijms24054601. Int J Mol Sci. 2023. PMID: 36902032 Free PMC article. Review.
-
Identification of key genes in periodontitis.Front Genet. 2025 Mar 19;16:1579848. doi: 10.3389/fgene.2025.1579848. eCollection 2025. Front Genet. 2025. PMID: 40176796 Free PMC article.
-
A new nano approach to prevent tumor growth in the local treatment of glioblastoma: Temozolomide and rutin-loaded hybrid layered composite nanofiber.Asian J Pharm Sci. 2024 Dec;19(6):100971. doi: 10.1016/j.ajps.2024.100971. Epub 2024 Oct 23. Asian J Pharm Sci. 2024. PMID: 39640055 Free PMC article.
-
Fine-scale mapping of chromosome 9q22.33 identifies candidate causal variant in ovarian cancer.PeerJ. 2024 Feb 14;12:e16918. doi: 10.7717/peerj.16918. eCollection 2024. PeerJ. 2024. PMID: 38371376 Free PMC article.
References
-
- Cedeno J.D., Light D.E., Leslie S.W. StatPearls. StatPearls Publishing; 2020. Testicular Seminoma; pp. 1–2.
-
- Siegel R.L., Miller K.D., Jemal A. Cancer statistics, 2020. CA Cancer J. Clin. 2020;70:7–30. - PubMed
-
- Bray F., Richiardi L., Ekbom A., Pukkala E., Cuninkova M., Møller H. Trends in testicular cancer incidence and mortality in 22 European countries: continuing increases in incidence and declines in mortality. Int. J. Cancer. 2006;118:3099–3111. - PubMed
-
- Znaor A., Lortet-Tieulent J., Jemal A., Bray F. International variations and trends in testicular cancer incidence and mortality. Eur. Urol. 2014;65:1095–1106. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

