Silencing long ascending propriospinal neurons after spinal cord injury improves hindlimb stepping in the adult rat
- PMID: 34854375
- PMCID: PMC8639151
- DOI: 10.7554/eLife.70058
Silencing long ascending propriospinal neurons after spinal cord injury improves hindlimb stepping in the adult rat
Abstract
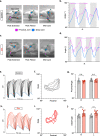
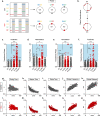
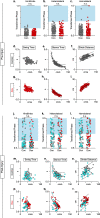
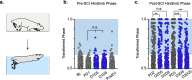
Long ascending propriospinal neurons (LAPNs) are a subpopulation of spinal cord interneurons that directly connect the lumbar and cervical enlargements. Previously we showed, in uninjured animals, that conditionally silencing LAPNs disrupted left-right coordination of the hindlimbs and forelimbs in a context-dependent manner, demonstrating that LAPNs secure alternation of the fore- and hindlimb pairs during overground stepping. Given the ventrolateral location of LAPN axons in the spinal cord white matter, many likely remain intact following incomplete, contusive, thoracic spinal cord injury (SCI), suggesting a potential role in the recovery of stepping. Thus, we hypothesized that silencing LAPNs after SCI would disrupt recovered locomotion. Instead, we found that silencing spared LAPNs post-SCI improved locomotor function, including paw placement order and timing, and a decrease in the number of dorsal steps. Silencing also restored left-right hindlimb coordination and normalized spatiotemporal features of gait such as stance and swing time. However, hindlimb-forelimb coordination was not restored. These data indicate that the temporal information carried between the spinal enlargements by the spared LAPNs post-SCI is detrimental to recovered hindlimb locomotor function. These findings are an illustration of a post-SCI neuroanatomical-functional paradox and have implications for the development of neuronal- and axonal-protective therapeutic strategies and the clinical study/implementation of neuromodulation strategies.
Keywords: locomotion; neuronal silencing; neuroscience; propriospinal neurons; rat; spinal cord injury; viral vector.
© 2021, Shepard et al.
Conflict of interest statement
CS, AP, BB, MV, RZ, DB, JM, AR, SW, DM No competing interests declared
Figures






References
-
- Arber T, Ruffion A, Terrier JE, Paparel P, Morel Journel N, Champetier D, Dominique I. Efficacy and security of continent catheterizable channels at short and middle term for adult neurogenic bladder dysfunction. Progres En Urologie. 2019;29:1047–1053. doi: 10.1016/j.purol.2019.08.278. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

