DNA damage response at telomeres boosts the transcription of SARS-CoV-2 receptor ACE2 during aging
- PMID: 34854526
- PMCID: PMC8811650
- DOI: 10.15252/embr.202153658
DNA damage response at telomeres boosts the transcription of SARS-CoV-2 receptor ACE2 during aging
Abstract
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes the coronavirus disease 2019 (COVID-19), known to be more common in the elderly, who also show more severe symptoms and are at higher risk of hospitalization and death. Here, we show that the expression of the angiotensin converting enzyme 2 (ACE2), the SARS-CoV-2 cell receptor, increases during aging in mouse and human lungs. ACE2 expression increases upon telomere shortening or dysfunction in both cultured mammalian cells and in vivo in mice. This increase is controlled at the transcriptional level, and Ace2 promoter activity is DNA damage response (DDR)-dependent. Both pharmacological global DDR inhibition of ATM kinase activity and selective telomeric DDR inhibition by the use of antisense oligonucleotides prevent Ace2 upregulation following telomere damage in cultured cells and in mice. We propose that during aging telomere dysfunction due to telomeric shortening or damage triggers DDR activation and this causes the upregulation of ACE2, the SARS-CoV-2 cell receptor, thus contributing to make the elderly more susceptible to the infection.
Keywords: Ace2; COVID-19; DNA damage response; aging; telomere.
© 2021 The Authors. Published under the terms of the CC BY NC ND 4.0 license.
Conflict of interest statement
F.R. and F.d’A.d.F. are inventors on the patent applications PCT/EP2013/059753 and PCT/EP2016/068162.
Figures

RT‐qPCR detection of Ace2 mRNA expression levels in lungs from young (2–3 months) and old (22–24 months) mice (n = 7 mice per group).
Representative microphotographs and quantitative analyses of ACE2 immunohistochemical staining in lungs from young (2 months) and old (22 months) mice (n = 3 mice per group). Scale bar, 200 µm.
Representative microphotographs and quantitative analyses of ACE2 immunohistochemical staining in lung parenchyma of young (20–35 years old) and old (60–80 years old) humans (n = 4–7 individuals per group). Scale bar, 200 µm.
Double‐marker immunofluorescence and quantitative analyses of ACE2 intensity level in pro‐SP‐C‐positive type II pneumocytic and CD31‐positive endothelia in lungs from young and old mice (n = 3–6 mice per group). Scale bar, 100 µm. a.u. = arbitrary units.
Double‐marker immunofluorescence and quantitative analyses of ACE2 intensity level in TTF‐1‐positive type II pneumocytic and CD31‐positive endothelia in lungs from young and old humans (n = 4–7 individuals per group). Scale bar, 100 µm. a.u. = arbitrary units.
Single‐cell transcriptomic data from aging tissues in the mouse lung from Tabula Muris Senis. Upper left panel: uniform manifold approximation and projection (UMAP) of cell identity with ATII cells represented as red dots. Upper right panel: dot plot of Ace2 expression according to age (young = 3 months; old = 30 months). Dot size represents fraction of cells expressing a given gene. Lower panel: UMAP of Ace2 expressing cells depicted as blue dots.

Quantitative analysis of ACE2 intensity level in pro‐SP‐C‐negative cells in lungs from young and old mice (n = 3 mice per group).
Quantitative analysis of ACE2 intensity level in TTF‐1‐negative cells in lungs from young and old humans (n = 4–7 individuals per group).
Single‐cell transcriptomic data in the mouse lung from Tabula Muris Senis. Left panel: UMAP of Gapdh expressing cells depicted as blue dots, with increasing color gradient according to expression intensity. Right panel: dot plot of Ace2 and Gapdh expression according to age (young = 3 months; old = 30 months). Dot size represents fraction of cells expressing a given gene.
Boxplot of Ace2 expression levels in type II pneumocytes of young (3 months) and old (30 months) mice from single‐cell transcriptomic data of aging tissues in the mouse lung from Tabula Muris Senis. The central dashed line, box edges, and whiskers indicate, respectively, the mean, upper and lower quartiles, and upper and lower fences.
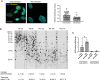
Representative image and quantification of the FISH signal using a telomeric PNA probe in early (PD 36.5) and late (PD 62) passage BJ. Scale bar, 20 µm. a.u. = arbitrary units.
The average and shortest telomere distribution in HBEC at the indicated PDs, as detected by Telomere Shortest Length Assay (TeSLA).
RT‐qPCR detection of ACE2 mRNA expression levels in early passages (PD 35) and late passages (PD 61–64) BJ, using primers specific for the long and short isoform (n = 3–4 replicates per group).
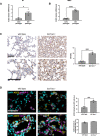
RT‐qPCR detection of ACE2 mRNA expression levels in early passages (PD 34–37) and late passages (PD 61‐64) human normal fibroblasts (BJ) (n = 3–4 replicates per group).
ddPCR detection of ACE2 mRNA expression levels in early passages (PD 22.37) and late passages (PD 140) human bronchial epithelial cells (HBEC) (n = 6 replicates per group).
Representative images and quantitative analyses of ACE2 immunohistochemical staining in lungs of age‐matched wild‐type and G3 Terc−/− mice (n = 3–4 mice per group). Scale bar, 200 µm.
Double‐marker immunofluorescence and quantitative analyses of ACE2 intensity level in pro‐SP‐C‐positive type II pneumocytic and CD31‐positive endothelia in lungs of age‐matched wild‐type and G3 Terc−/− mice (n = 3–4 mice per group). Scale bar, 100 µm. a.u. = arbitrary units.

Representative image and quantification of the FISH signal using a telomeric PNA probe in the lungs from wild‐type or G3 Terc−/− mice. Scale bar, 10 µm. a.u. = arbitrary units (n = 3–4 mice per group).
Quantitative analysis of ACE2 intensity level in pro‐SP‐C‐negative cells in lungs from wild‐type or G3 Terc−/− mice. a.u. = arbitrary units (n = 3–4 mice per group).
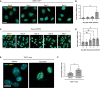
Immunofluorescence showing γH2AX foci in MEFs Trf2F/F at the indicated time points following 4OHT treatment and consequent TRF2 knockout. Scale bar, 25 µm.
RT‐qPCR detection of Ace2 mRNA expression levels in MEFs Trf2F/F treated as in A (n = 3 independent experiments).
Immunofluorescence showing γH2AX foci in HeLa shTRF2 cells at the indicated time points following doxycycline treatment and consequent TRF2 knockdown. Scale bar, 25 µm.
RT‐qPCR detection of ACE2 mRNA expression levels in HeLa shTRF2 cells treated as in C (n = 6 independent experiments).
Representative immunofluorescence images of 53BP1 staining in liver from Trf2F/F mice treated with tamoxifen (to induce TRF2 loss and telomere uncapping) or vehicle. The animals have been injected also with PBS. Scale bar, 10 µm.
RT‐qPCR detection of Ace2 mRNA expression levels in livers of mice treated as in E (n = 5–7 mice per group).
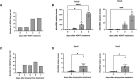
Quantification of the number of γH2AX foci per cell related to Fig 3A.
RT‐qPCR detection of telomeric dilncRNA expression levels in MEFs Trf2F/F following 4OHT treatment (n = 3 independent experiments).
Quantification of the number of γH2AX foci per cell related to Fig 3C.
RT‐qPCR detection of telomeric dilncRNA expression levels in HeLa shTRF2 following doxycycline treatment (n = 6 independent experiments).
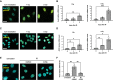
Immunofluorescence showing γH2AX foci 6 h after the indicated dose of ionizing radiation in MEFs Trf2F/F. Scale bar, 25 µm.
RT‐qPCR detection of Ace2 expression levels in MEFs Trf2F/F treated as in A (n = 3 independent experiments).
Immunofluorescence showing γH2AX foci 6 h after the indicated dose of ionizing radiation in HeLa shTRF2. Scale bar, 25 µm.
RT‐qPCR detection of ACE2 expression levels in HeLa shTRF2 treated as in C (n = 3 independent experiments).
Immunofluorescence showing γH2AX foci 6 h after 5 Gy of ionizing radiation in MEFs Trf2F/F, treated with DMSO or ATMi. Scale bar 25 µm.
RT‐qPCR detection of Ace2 expression levels in MEFs Trf2F/F treated as in E (n = 3 independent experiments).
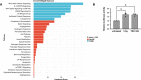
Gene set enrichment analysis showing enriched pathways from the MSigDB_Hallmark gene set library. Bars are colored according to P‐value, with significantly enriched pathways (P < 0.05) in blue. Combined score: combined value of both the P‐value and z‐score.
Relative luciferase activity in HeLa shTRF2 following ionizing radiation (5 Gy) or TRF2 knockdown upon doxycycline‐induced shTRF2 expression (n = 3 independent experiments). Error bars represent the SEM. *P < 0.05. Two‐way paired ANOVA.
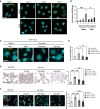
Immunofluorescence showing γH2AX foci in Trf2F/F MEFs at the indicated time points following 4OHT treatment and consequent TRF2 knockout and treated with DMSO or ATMi. Scale bar, 25 µm.
RT‐qPCR detection of Ace2 mRNA expression levels in MEFs Trf2F/F treated as in A (n = 3 independent experiments).
Representative immunofluorescence images of 53BP1 staining in liver from Trf2F/F mice treated with tamoxifen (to induce telomere uncapping) or vehicle and injected with the indicated ASOs or PBS as control. Scale bar, 10 µm.
RT‐qPCR detection of Ace2 mRNA levels in livers of mice treated as in C (n = 5–8 mice per group).
Representative microphotographs and quantitative analyses of ACE2 immunohistochemical staining in lungs of age‐matched wild‐type and G3 Terc−/− mice, treated with the indicated ASOs (n = 4–9 mice per group). Scale bar, 200 µm.
Double‐marker immunofluorescence and quantitative analyses of ACE2 intensity level in pro‐SP‐C‐positive type II pneumocytic cells in lungs of age‐matched wild‐type and G3 Terc−/− mice, treated with the indicated ASOs (n = 4–9 mice per group). Scale bar, 100 µm. a.u. = arbitrary units.
Similar articles
-
DNA damage contributes to age-associated differences in SARS-CoV-2 infection.Aging Cell. 2022 Dec;21(12):e13729. doi: 10.1111/acel.13729. Epub 2022 Oct 18. Aging Cell. 2022. PMID: 36254583 Free PMC article.
-
Increasing host cellular receptor-angiotensin-converting enzyme 2 expression by coronavirus may facilitate 2019-nCoV (or SARS-CoV-2) infection.J Med Virol. 2020 Nov;92(11):2693-2701. doi: 10.1002/jmv.26139. Epub 2020 Jul 2. J Med Virol. 2020. PMID: 32497323 Free PMC article.
-
SARS-CoV-2 Causes Lung Infection without Severe Disease in Human ACE2 Knock-In Mice.J Virol. 2022 Jan 12;96(1):e0151121. doi: 10.1128/JVI.01511-21. Epub 2021 Oct 20. J Virol. 2022. PMID: 34668780 Free PMC article.
-
Angiotensin-converting enzyme 2 (ACE2), SARS-CoV-2 and the pathophysiology of coronavirus disease 2019 (COVID-19).J Pathol. 2020 Jul;251(3):228-248. doi: 10.1002/path.5471. Epub 2020 Jun 10. J Pathol. 2020. PMID: 32418199 Free PMC article. Review.
-
SARS-CoV-2 pandemic and research gaps: Understanding SARS-CoV-2 interaction with the ACE2 receptor and implications for therapy.Theranostics. 2020 Jun 12;10(16):7448-7464. doi: 10.7150/thno.48076. eCollection 2020. Theranostics. 2020. PMID: 32642005 Free PMC article. Review.
Cited by
-
Roles of chromatin and genome instability in cellular senescence and their relevance to ageing and related diseases.Nat Rev Mol Cell Biol. 2024 Dec;25(12):979-1000. doi: 10.1038/s41580-024-00775-3. Epub 2024 Oct 3. Nat Rev Mol Cell Biol. 2024. PMID: 39363000 Review.
-
Self-DNA driven inflammation in COVID-19 and after mRNA-based vaccination: lessons for non-COVID-19 pathologies.Front Immunol. 2024 Feb 19;14:1259879. doi: 10.3389/fimmu.2023.1259879. eCollection 2023. Front Immunol. 2024. PMID: 38439942 Free PMC article.
-
Radiotherapy for non-cancer diseases: benefits and long-term risks.Int J Radiat Biol. 2024;100(4):505-526. doi: 10.1080/09553002.2023.2295966. Epub 2024 Jan 5. Int J Radiat Biol. 2024. PMID: 38180039 Free PMC article. Review.
-
ABO Blood System and COVID-19 Susceptibility: Anti-A and Anti-B Antibodies Are the Key Points.Front Med (Lausanne). 2022 Apr 25;9:882477. doi: 10.3389/fmed.2022.882477. eCollection 2022. Front Med (Lausanne). 2022. PMID: 35547235 Free PMC article. Review.
-
Factors Modulating COVID-19: A Mechanistic Understanding Based on the Adverse Outcome Pathway Framework.J Clin Med. 2022 Jul 31;11(15):4464. doi: 10.3390/jcm11154464. J Clin Med. 2022. PMID: 35956081 Free PMC article. Review.
References
-
- Aguado J, Sola‐Carvajal A, Cancila V, Revêchon G, Ong PF, Jones‐Weinert CW, Wallén Arzt E, Lattanzi G, Dreesen O, Tripodo C et al (2019) Inhibition of DNA damage response at telomeres improves the detrimental phenotypes of Hutchinson‐Gilford Progeria Syndrome. Nat Commun 10: 4990–5000 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- Fondazione Umberto Veronesi (Umberto Veronesi Foundation)
- 835103/EC|H2020|H2020 Priority Excellent Science|H2020 European Research Council (ERC)
- 875139/EC|H2020|H2020 Priority Excellent Science|H2020 European Research Council (ERC)
- 21091/Associazione Italiana per la Ricerca sul Cancro (AIRC)
- P50 CA070907/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

