Pharmacokinetics of high-titer anti-SARS-CoV-2 human convalescent plasma in high-risk children
- PMID: 34855624
- PMCID: PMC8855821
- DOI: 10.1172/jci.insight.151518
Pharmacokinetics of high-titer anti-SARS-CoV-2 human convalescent plasma in high-risk children
Abstract
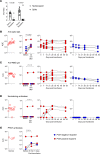
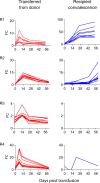
BACKGROUNDWhile most children who contract COVID-19 experience mild disease, high-risk children with underlying conditions may develop severe disease, requiring interventions. Kinetics of antibodies transferred via COVID-19 convalescent plasma early in disease have not been characterized.METHODSIn this study, high-risk children were prospectively enrolled to receive high-titer COVID-19 convalescent plasma (>1:320 anti-spike IgG; Euroimmun). Passive transfer of antibodies and endogenous antibody production were serially evaluated for up to 2 months after transfusion. Commercial and research ELISA assays, virus neutralization assays, high-throughput phage-display assay utilizing a coronavirus epitope library, and pharmacokinetic analyses were performed.RESULTSFourteen high-risk children (median age, 7.5 years) received high-titer COVID-19 convalescent plasma, 9 children within 5 days (range, 2-7 days) of symptom onset and 5 children within 4 days (range, 3-5 days) after exposure to SARS-CoV-2. There were no serious adverse events related to transfusion. Antibodies against SARS-CoV-2 were transferred from the donor to the recipient, but antibody titers declined by 14-21 days, with a 15.1-day half-life for spike protein IgG. Donor plasma had significant neutralization capacity, which was transferred to the recipient. However, as early as 30 minutes after transfusion, recipient plasma neutralization titers were 6.2% (range, 5.9%-6.7%) of donor titers.CONCLUSIONConvalescent plasma transfused to high-risk children appears to be safe, with expected antibody kinetics, regardless of weight or age. However, current use of convalescent plasma in high-risk children achieves neutralizing capacity, which may protect against severe disease but is unlikely to provide lasting protection.Trial registrationClinicalTrials.gov NCT04377672.FundingThe state of Maryland, Bloomberg Philanthropies, and the NIH (grants R01-AI153349, R01-AI145435-A1, K08-AI139371-A1, and T32-AI052071).
Keywords: COVID-19; Immunoglobulins; Infectious disease.
Conflict of interest statement
Figures


References
-
- Kimberlin DW, et al, eds. Red Book: 2018-2021 Report of the Committee on Infectious Diseases. AAP Publications; 2018.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

