The Dorsal Skinfold Chamber as a New Tympanic Membrane Wound Healing Model: Intravital Insights into the Pathophysiology of Epithelialized Wounds
- PMID: 34856545
- PMCID: PMC9808650
- DOI: 10.1159/000519774
The Dorsal Skinfold Chamber as a New Tympanic Membrane Wound Healing Model: Intravital Insights into the Pathophysiology of Epithelialized Wounds
Abstract
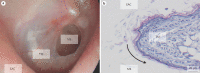
Background: Tympanic membrane perforations (TMPs) are a common complication of trauma and infection. Persisting perforations result from the unique location of the tympanic membrane. The wound is surrounded by air of the middle ear and the external auditory canal. The inadequate wound bed, growth factor, and blood supply lead to circular epithelialization of the perforation's edge and premature interruption of defect closure. Orthotopic animal models use mechanical or chemical tympanic membrane laceration to identify bioactive wound dressings and overcome premature epithelialization. However, all orthotopic models essentially lack repetitive visualization of the biomaterial-wound interface. Therefore, recent progress in 3D printing of customized wound dressings has not yet been transferred to the unique wound setup of the TMP. Here, we present a novel application for the mice dorsal skinfold chamber (DSC) with an epithelialized full-thickness defect as TMP model.
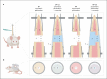
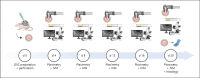
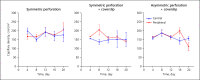
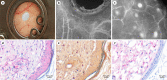
Methods: A circular 2-mm defect was cut into the extended dorsal skinfold using a biopsy punch. The skinfold was either perforated through both skin layers without prior preparation or perforated on 1 side, following resection of the opposing skin layer. In both groups, the wound was sealed with a coverslip or left unclosed (n = 4). All animals were examined for epithelialization of the edge (histology), size of the perforation (planimetry), neovascularization (repetitive intravital fluorescence microscopy), and inflammation (immunohistology).
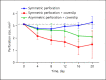
Results: The edge of the perforation was overgrown by the cornified squamous epithelium in all pre-parations. Reduction in the perforation's size was enhanced by application of a coverslip. Microsurgical preparation before biopsy punch perforation and sealing with a coverslip enabled repetitive high-quality intravital fluorescence microscopy. However, spontaneous reduction of the perforation occurred frequently. Therefore, the direct biopsy punch perforation without microsurgical preparation was favorable: spontaneous reduction did not occur throughout 21 days. Moreover, the visualization of the neovascularization was sufficient in intravital microscopy.
Conclusions: The DSC full-thickness defect is a valuable supplement to orthotopic TMP models. Repetitive intravital microscopy of the epithelialized edge enables investigation of the underlying pathophysiology during the transition from the inflammation to the proliferation phase of wound healing. Using established analysis procedures, the present model provides an effective platform for the screening of bioactive materials and transferring progress in tissue engineering to the special conditions of tympanic membrane wound healing.
Keywords: Biomaterials; Tympanic membrane perforation; Wound dressings; Wound healing models.
© 2021 The Author(s). Published by S. Karger AG, Basel.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Repair of Tympanic Membrane Perforations with Customized Bioprinted Ear Grafts Using Chinchilla Models.Tissue Eng Part A. 2018 Mar;24(5-6):527-535. doi: 10.1089/ten.TEA.2017.0246. Epub 2017 Sep 1. Tissue Eng Part A. 2018. PMID: 28726587 Free PMC article.
-
Rat model of chronic tympanic membrane perforation: A longitudinal histological evaluation of underlying mechanisms.Int J Pediatr Otorhinolaryngol. 2017 Feb;93:88-96. doi: 10.1016/j.ijporl.2016.12.028. Epub 2016 Dec 25. Int J Pediatr Otorhinolaryngol. 2017. PMID: 28109506
-
Animal Model of Chronic Tympanic Membrane Perforation.Anat Rec (Hoboken). 2020 Mar;303(3):619-625. doi: 10.1002/ar.24220. Epub 2019 Jul 16. Anat Rec (Hoboken). 2020. PMID: 31260172
-
Necessities, opportunities, and challenges for tympanic membrane perforation scaffolding-based bioengineering.Biomed Mater. 2021 Mar 3;16(3). doi: 10.1088/1748-605X/abcf5d. Biomed Mater. 2021. PMID: 33260166 Review.
-
A moist edge environment aids the regeneration of traumatic tympanic membrane perforations.J Laryngol Otol. 2017 Jul;131(7):564-571. doi: 10.1017/S0022215117001001. Epub 2017 May 15. J Laryngol Otol. 2017. PMID: 28502255 Review.
References
MeSH terms
LinkOut - more resources
Full Text Sources

