Hamster models of COVID-19 pneumonia reviewed: How human can they be?
- PMID: 34856819
- PMCID: PMC9207993
- DOI: 10.1177/03009858211057197
Hamster models of COVID-19 pneumonia reviewed: How human can they be?
Abstract
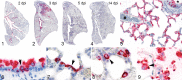
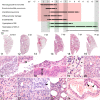
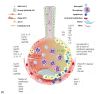
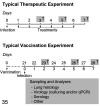
The dramatic global consequences of the coronavirus disease 2019 (COVID-19) pandemic soon fueled quests for a suitable model that would facilitate the development and testing of therapies and vaccines. In contrast to other rodents, hamsters are naturally susceptible to infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and the Syrian hamster (Mesocricetus auratus) rapidly developed into a popular model. It recapitulates many characteristic features as seen in patients with a moderate, self-limiting course of the disease such as specific patterns of respiratory tract inflammation, vascular endothelialitis, and age dependence. Among 4 other hamster species examined, the Roborovski dwarf hamster (Phodopus roborovskii) more closely mimics the disease in highly susceptible patients with frequent lethal outcome, including devastating diffuse alveolar damage and coagulopathy. Thus, different hamster species are available to mimic different courses of the wide spectrum of COVID-19 manifestations in humans. On the other hand, fewer diagnostic tools and information on immune functions and molecular pathways are available than in mice, which limits mechanistic studies and inference to humans in several aspects. Still, under pandemic conditions with high pressure on progress in both basic and clinically oriented research, the Syrian hamster has turned into the leading non-transgenic model at an unprecedented pace, currently used in innumerable studies that all aim to combat the impact of the virus with its new variants of concern. As in other models, its strength rests upon a solid understanding of its similarities to and differences from the human disease, which we review here.
Keywords: COVID-19; animal model; hamster; histopathology; pathology; preclinical research; review; severe acute respiratory syndrome coronavirus 2; vaccines.
Conflict of interest statement
Figures

Similar articles
-
The Roborovski Dwarf Hamster Is A Highly Susceptible Model for a Rapid and Fatal Course of SARS-CoV-2 Infection.Cell Rep. 2020 Dec 8;33(10):108488. doi: 10.1016/j.celrep.2020.108488. Epub 2020 Nov 19. Cell Rep. 2020. PMID: 33271063 Free PMC article.
-
De Novo-Whole Genome Assembly of the Roborovski Dwarf Hamster (Phodopus roborovskii) Genome: An Animal Model for Severe/Critical COVID-19.Genome Biol Evol. 2022 Jul 2;14(7):evac100. doi: 10.1093/gbe/evac100. Genome Biol Evol. 2022. PMID: 35778793 Free PMC article.
-
Roborovski hamster (Phodopus roborovskii) strain SH101 as a systemic infection model of SARS-CoV-2.Virulence. 2021 Dec;12(1):2430-2442. doi: 10.1080/21505594.2021.1972201. Virulence. 2021. PMID: 34517779 Free PMC article.
-
Hamsters as a Model of Severe Acute Respiratory Syndrome Coronavirus-2.Comp Med. 2021 Oct 1;71(5):398-410. doi: 10.30802/AALAS-CM-21-000036. Epub 2021 Sep 29. Comp Med. 2021. PMID: 34588095 Free PMC article. Review.
-
[Susceptibility of animal species to experimental SARS-CoV-2 (Coronaviridae: Coronavirinae: Betacoronavirus; Sarbecovirus) infection].Vopr Virusol. 2021 May 15;66(2):103-111. doi: 10.36233/0507-4088-47. Vopr Virusol. 2021. PMID: 33993680 Review. Russian.
Cited by
-
Immunobiology of COVID-19: Mechanistic and therapeutic insights from animal models.Zool Res. 2024 Jul 18;45(4):747-766. doi: 10.24272/j.issn.2095-8137.2024.062. Zool Res. 2024. PMID: 38894519 Free PMC article. Review.
-
Impaired inflammatory resolution with severe SARS-CoV-2 infection in leptin knock out obese hamster.iScience. 2025 Jan 17;28(2):111837. doi: 10.1016/j.isci.2025.111837. eCollection 2025 Feb 21. iScience. 2025. PMID: 39981511 Free PMC article.
-
Rehoming and Other Refinements and Replacement in Procedures Using Golden Hamsters in SARS-CoV-2 Vaccine Research.Animals (Basel). 2023 Aug 14;13(16):2616. doi: 10.3390/ani13162616. Animals (Basel). 2023. PMID: 37627407 Free PMC article.
-
Differential patterns of cross-protection against antigenically distinct variants in small animal models of SARS-CoV-2 infection.Npj Viruses. 2025 Jun 4;3(1):49. doi: 10.1038/s44298-025-00125-w. Npj Viruses. 2025. PMID: 40467943 Free PMC article.
-
Investigation of SARS-CoV-2 individual proteins reveals the in vitro and in vivo immunogenicity of membrane protein.Sci Rep. 2023 Dec 18;13(1):22873. doi: 10.1038/s41598-023-49077-2. Sci Rep. 2023. PMID: 38129491 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

