Ameliorating Alzheimer's-like Pathology by Minocycline via Inhibiting Cdk5/p25 Signaling
- PMID: 34856907
- PMCID: PMC9881058
- DOI: 10.2174/1570159X19666211202124925
Ameliorating Alzheimer's-like Pathology by Minocycline via Inhibiting Cdk5/p25 Signaling
Abstract
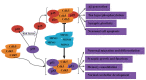
Background: Minocycline has multiple neuroprotective roles in abundant brain diseases, including the prevention and treatment of Alzheimer's disease (AD). Cdk5/p25 signaling plays an important role in the onset and development of Alzheimer's-like pathology. The aim of the present work was to further explore the underlying mechanism which minocycline effects on Cdk5/p25 signaling related to Alzheimer's-like pathology.
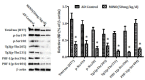
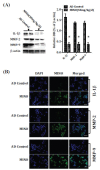
Methods: The cognitive function of animals was measured by the Morris water maze test. The levels of Aβ were determined by an enzyme-linked immunosorbent assay. The levels of APP, β- and γ- secretases, and the biomarkers of tau (total tau and hyperphosphorylated tau), inflammatory cytokine and matrix metalloproteinases (MMP-2 and MMP-9), and biomarkers of synapse and Cdk5/p25 signaling, were detected by the Western blotting. The biomarkers of the synapse, inflammatory cytokine, and matrix metalloproteinases (MMP-2 and MMP-9) were also determined by immunofluorescence.
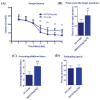
Results: Minocycline improved learning and memory in APP/PS1 mice. It limited the production of Aβ and hyperphosphorylation of tau in the hippocampus and ameliorated synaptic deficit. Moreover, it also inhibited the activation of Cdk5/p25 signaling, inflammation, and matrix metalloproteinases.
Conclusion: Minocycline mitigates Alzheimer's-like pathology via limiting the activation of Cdk5/p25 signaling pathway and improves cognitive deficits.
Keywords: Alzheimer’s disease; Minocycline; anti-inflammatory drugs; cyclin-dependent kinase 5; pathology; synapse.
Copyright© Bentham Science Publishers; For any queries, please email at epub@benthamscience.net.
Figures





Similar articles
-
Sulforaphene, a CDK5 Inhibitor, attenuates cognitive deficits in a transgenic mouse model of Alzheimer's disease via reducing Aβ Deposition, tau hyperphosphorylation and synaptic dysfunction.Int Immunopharmacol. 2023 Jan;114:109504. doi: 10.1016/j.intimp.2022.109504. Epub 2022 Dec 9. Int Immunopharmacol. 2023. PMID: 36508924
-
Synaptic deficits are rescued in the p25/Cdk5 model of neurodegeneration by the reduction of β-secretase (BACE1).J Neurosci. 2011 Nov 2;31(44):15751-6. doi: 10.1523/JNEUROSCI.3588-11.2011. J Neurosci. 2011. PMID: 22049418 Free PMC article.
-
Hypoxia increases Aβ-induced tau phosphorylation by calpain and promotes behavioral consequences in AD transgenic mice.J Mol Neurosci. 2013 Sep;51(1):138-47. doi: 10.1007/s12031-013-9966-y. Epub 2013 Jan 24. J Mol Neurosci. 2013. PMID: 23345083
-
Deregulated Cdk5 activity is involved in inducing Alzheimer's disease.Arch Med Res. 2012 Nov;43(8):655-62. doi: 10.1016/j.arcmed.2012.10.015. Epub 2012 Nov 7. Arch Med Res. 2012. PMID: 23142263 Free PMC article. Review.
-
Cdk5-p25 as a key element linking amyloid and tau pathologies in Alzheimer's disease: Mechanisms and possible therapeutic interventions.Life Sci. 2022 Nov 1;308:120986. doi: 10.1016/j.lfs.2022.120986. Epub 2022 Sep 21. Life Sci. 2022. PMID: 36152679 Review.
Cited by
-
Focusing on cyclin-dependent kinases 5: A potential target for neurological disorders.Front Mol Neurosci. 2022 Nov 10;15:1030639. doi: 10.3389/fnmol.2022.1030639. eCollection 2022. Front Mol Neurosci. 2022. PMID: 36438186 Free PMC article. Review.
-
Neuroinflammation mediates the progression of neonate hypoxia-ischemia brain damage to Alzheimer's disease: a bioinformatics and experimental study.Front Aging Neurosci. 2025 Jan 13;16:1511668. doi: 10.3389/fnagi.2024.1511668. eCollection 2024. Front Aging Neurosci. 2025. PMID: 39872979 Free PMC article.
-
Systemic inflammation as a central player in the initiation and development of Alzheimer's disease.Immun Ageing. 2025 Aug 21;22(1):33. doi: 10.1186/s12979-025-00529-5. Immun Ageing. 2025. PMID: 40841660 Free PMC article. Review.
-
Newer Therapeutic Approaches in Treating Alzheimer's Disease: A Comprehensive Review.ACS Omega. 2025 Feb 3;10(6):5148-5171. doi: 10.1021/acsomega.4c05527. eCollection 2025 Feb 18. ACS Omega. 2025. PMID: 39989768 Free PMC article. Review.
-
Minocycline as a prospective therapeutic agent for cancer and non-cancer diseases: a scoping review.Naunyn Schmiedebergs Arch Pharmacol. 2024 May;397(5):2835-2848. doi: 10.1007/s00210-023-02839-1. Epub 2023 Nov 22. Naunyn Schmiedebergs Arch Pharmacol. 2024. PMID: 37991540
References
-
- Blautzik J., Keeser D., Paolini M., Kirsch V., Berman A., Coates U., Reiser M., Teipel S.J., Meindl T. Functional connectivity increase in the default-mode network of patients with Alzheimer’s disease after long-term treatment with Galantamine. Eur. Neuropsychopharmacol. 2016;26(3):602–613. doi: 10.1016/j.euroneuro.2015.12.006. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

