Phase 2 Trial of Rituximab in Acetylcholine Receptor Antibody-Positive Generalized Myasthenia Gravis: The BeatMG Study
- PMID: 34857535
- PMCID: PMC8793103
- DOI: 10.1212/WNL.0000000000013121
Phase 2 Trial of Rituximab in Acetylcholine Receptor Antibody-Positive Generalized Myasthenia Gravis: The BeatMG Study
Abstract
Objective: To determine whether rituximab is safe and potentially beneficial, warranting further investigation in an efficacy trial for acetylcholine receptor antibody-positive generalized MG (AChR-Ab+ gMG).
Methods:
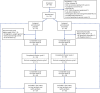
The B-Cell Targeted Treatment in MG (BeatMG) study was a randomized, double-blind, placebo-controlled, multicenter phase-2 trial that utilized a futility design. Individuals 21-90 years of age, with AChR-Ab+ gMG (MG Foundation of America Class II-IV) and receiving prednisone ≥15 mg/day were eligible. The primary outcome was a measure of steroid-sparing effect, defined as the proportion achieving ≥75% reduction in mean daily prednisone dose in the 4-weeks prior to week 52
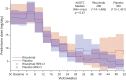
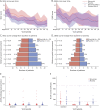
Results: Of the 52 participants included, mean (±SD) age at enrollment was 55.1 (±17.1) years; 23 (44.2%) were female, and 31 (59.6%) were MGFA Class II. The mean (±SD) baseline prednisone dose was 22.1 (±9.7) mg/day. The primary steroid-sparing outcome was achieved in 60% of those on rituximab vs. 56% on placebo. The study reached its futility endpoint (p=0.03) suggesting that the pre-defined clinically meaningful improvement of 30% due to rituximab over placebo was unlikely to be achieved in a subsequent, larger trial. No safety issues identified.
Conclusions: While rituximab was safe and well-tolerated, these results suggest that there is a low probability of observing the defined clinically meaningful steroid-sparing effect over a 12-month period in a phase-3 trial of mild-moderately symptomatic AChR-Ab+ gMG.
Classification of evidence: This study provides Class I evidence that for mild-to-moderate AChR-Ab+ gMG, compared with placebo, rituximab is safe but unlikely to reduce steroid use by an absolute difference of at least 30% at 1 year.
Trial registration: ClinicalTrials.gov Identifier: NCT02110706.
© 2021 American Academy of Neurology.
Figures
References
-
- Dalakas MC. Invited article: inhibition of B cell functions: implications for neurology. Neurology. 2008;70(23):2252-2260. - PubMed
-
- Lebrun C, Bourg V, Tieulie N, Thomas P. Successful treatment of refractory generalized myasthenia gravis with rituximab. Eur J Neurol. 2009;16(2):246-250. - PubMed
-
- Diaz-Manera J, Martinez-Hernandez E, Querol L, et al. Long-lasting treatment effect of rituximab in MuSK myasthenia. Neurology. 2012;78(3):189-193. - PubMed
-
- Illa I, Diaz-Manera J, Rojas-Garcia R, et al. Sustained response to Rituximab in anti-AChR and anti-MuSK positive Myasthenia Gravis patients. J Neuroimmunol. 2008;201-202:90-94. - PubMed
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
