POSNOC-POsitive Sentinel NOde: adjuvant therapy alone versus adjuvant therapy plus Clearance or axillary radiotherapy: a randomised controlled trial of axillary treatment in women with early-stage breast cancer who have metastases in one or two sentinel nodes
- PMID: 34857578
- PMCID: PMC8640630
- DOI: 10.1136/bmjopen-2021-054365
POSNOC-POsitive Sentinel NOde: adjuvant therapy alone versus adjuvant therapy plus Clearance or axillary radiotherapy: a randomised controlled trial of axillary treatment in women with early-stage breast cancer who have metastases in one or two sentinel nodes
Abstract
Introduction: ACOSOG-Z0011(Z11) trial showed that axillary node clearance (ANC) may be omitted in women with ≤2 positive nodes undergoing breast conserving surgery (BCS) and whole breast radiotherapy (RT). A confirmatory study is needed to clarify the role of axillary treatment in women with ≤2 macrometastases undergoing BCS and groups that were not included in Z11 for example, mastectomy and those with microscopic extranodal invasion. The primary objective of POsitive Sentinel NOde: adjuvant therapy alone versus adjuvant therapy plus Clearance or axillary radiotherapy (POSNOC) is to evaluate whether for women with breast cancer and 1 or 2 macrometastases, adjuvant therapy alone is non-inferior to adjuvant therapy plus axillary treatment, in terms of 5-year axillary recurrence.
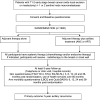
Methods and analysis: POSNOC is a pragmatic, multicentre, non-inferiority, international trial with participants randomised in a 1:1 ratio. Women are eligible if they have T1/T2, unifocal or multifocal invasive breast cancer, and 1 or 2 macrometastases at sentinel node biopsy, with or without extranodal extension. In the intervention group women receive adjuvant therapy alone, in the standard care group they receive ANC or axillary RT. In both groups women receive adjuvant therapy, according to local guidelines. This includes systemic therapy and, if indicated, RT to breast or chest wall. The UK Radiotherapy Trials Quality Assurance Group manages the in-built radiotherapy quality assurance programme. Primary endpoint is 5-year axillary recurrence. Secondary outcomes are arm morbidity assessed by Lymphoedema and Breast Cancer Questionnaire and QuickDASH questionnaires; quality of life and anxiety as assessed with FACT B+4 and State/Trait Anxiety Inventory questionnaires, respectively; other oncological outcomes; economic evaluation using EQ-5D-5L. Target sample size is 1900. Primary analysis is per protocol. Recruitment started on 1 August 2014 and as of 9 June 2021, 1866 participants have been randomised.
Ethics and dissemination: Protocol was approved by the National Research Ethics Service Committee East Midlands-Nottingham 2 (REC reference: 13/EM/0459). Results will be submitted for publication in peer-reviewed journals.
Trial registration number: ISRCTN54765244; NCT0240168Cite Now.
Keywords: adult radiotherapy; breast surgery; breast tumours.
© Author(s) (or their employer(s)) 2021. Re-use permitted under CC BY. Published by BMJ.
Conflict of interest statement
Competing interests: LV was a member of the NIHR Health Technology Assessment Programme, Clinical Trials and Evaluation Panel from 2014 to 2018.
Figures
References
-
- Giuliano AE, McCall L, Beitsch P, et al. . Locoregional recurrence after sentinel lymph node dissection with or without axillary dissection in patients with sentinel lymph node metastases: the American College of surgeons Oncology Group Z0011 randomized trial. Ann Surg 2010;252:426–32. 10.1097/SLA.0b013e3181f08f32 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
