SARS-CoV-2 spike protein induces inflammation via TLR2-dependent activation of the NF-κB pathway
- PMID: 34866574
- PMCID: PMC8709575
- DOI: 10.7554/eLife.68563
SARS-CoV-2 spike protein induces inflammation via TLR2-dependent activation of the NF-κB pathway
Abstract
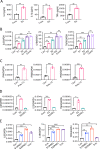
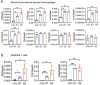
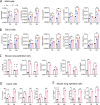
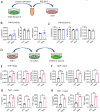
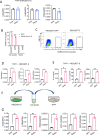
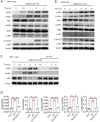
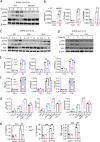
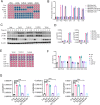
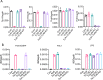
The pathogenesis of COVID-19 is associated with a hyperinflammatory response; however, the precise mechanism of SARS-CoV-2-induced inflammation is poorly understood. Here, we investigated direct inflammatory functions of major structural proteins of SARS-CoV-2. We observed that spike (S) protein potently induced inflammatory cytokines and chemokines, including IL-6, IL-1β, TNFα, CXCL1, CXCL2, and CCL2, but not IFNs in human and mouse macrophages. No such inflammatory response was observed in response to membrane (M), envelope (E), and nucleocapsid (N) proteins. When stimulated with extracellular S protein, human and mouse lung epithelial cells also produced inflammatory cytokines and chemokines. Interestingly, epithelial cells expressing S protein intracellularly were non-inflammatory, but elicited an inflammatory response in macrophages when co-cultured. Biochemical studies revealed that S protein triggers inflammation via activation of the NF-κB pathway in a MyD88-dependent manner. Further, such an activation of the NF-κB pathway was abrogated in Tlr2-deficient macrophages. Consistently, administration of S protein-induced IL-6, TNF-α, and IL-1β in wild-type, but not Tlr2-deficient mice. Notably, upon recognition of S protein, TLR2 dimerizes with TLR1 or TLR6 to activate the NF-κB pathway. Taken together, these data reveal a mechanism for the cytokine storm during SARS-CoV-2 infection and suggest that TLR2 could be a potential therapeutic target for COVID-19.
Keywords: COVID-19; SARS-CoV-2; TLR2; cytokine storm; immunology; inflammation; spike protein; viruses.
© 2021, Khan et al.
Conflict of interest statement
SK, MS, CL, RS, HZ No competing interests declared, JS Reviewing editor, eLife
Figures

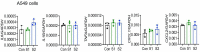


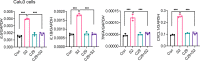

Update of
-
SARS-CoV-2 spike protein induces inflammation via TLR2-dependent activation of the NF-κB pathway.bioRxiv [Preprint]. 2021 Mar 17:2021.03.16.435700. doi: 10.1101/2021.03.16.435700. bioRxiv. 2021. Update in: Elife. 2021 Dec 06;10:e68563. doi: 10.7554/eLife.68563. PMID: 33758854 Free PMC article. Updated. Preprint.
References
-
- Bertram S, Glowacka I, Müller MA, Lavender H, Gnirss K, Nehlmeier I, Niemeyer D, He Y, Simmons G, Drosten C, Soilleux EJ, Jahn O, Steffen I, Pöhlmann S. Cleavage and activation of the severe acute respiratory syndrome coronavirus spike protein by human airway trypsin-like protease. Journal of Virology. 2011;85:13363–13372. doi: 10.1128/JVI.05300-11. - DOI - PMC - PubMed
-
- Blanco-Melo D, Nilsson-Payant BE, Liu W-C, Uhl S, Hoagland D, Møller R, Jordan TX, Oishi K, Panis M, Sachs D, Wang TT, Schwartz RE, Lim JK, Albrecht RA, tenOever BR. Imbalanced Host Response to SARS-CoV-2 Drives Development of COVID-19. Cell. 2020;181:1036–1045. doi: 10.1016/j.cell.2020.04.026. - DOI - PMC - PubMed
-
- Chang Y-J, Liu CY-Y, Chiang B-L, Chao Y-C, Chen C-C. Induction of IL-8 release in lung cells via activator protein-1 by recombinant baculovirus displaying severe acute respiratory syndrome-coronavirus spike proteins: identification of two functional regions. Journal of Immunology. 2004;173:7602–7614. doi: 10.4049/jimmunol.173.12.7602. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

