Low-Dose Piperlongumine Rescues Impaired Function of Endothelial Progenitor Cells and Reduces Cerebral Ischemic Injury in High-Fat Diet-Fed Mice
- PMID: 34867315
- PMCID: PMC8634707
- DOI: 10.3389/fphar.2021.689880
Low-Dose Piperlongumine Rescues Impaired Function of Endothelial Progenitor Cells and Reduces Cerebral Ischemic Injury in High-Fat Diet-Fed Mice
Abstract
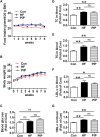
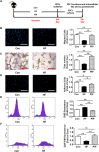
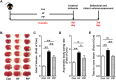
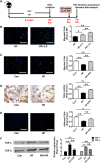
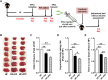
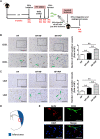
It is of great clinical significance to develop potential novel strategies to prevent cardio-cerebrovascular complications in patients with hyperlipidemia. Vascular Endothelial integrity and function play a key role in the prevention of cardio-cerebrovascular diseases. Endothelial progenitor cells (EPCs) can home to sites of ischemic injury and promote endothelial regeneration and neovascularization. Hypercholesterolemia impairs the function of EPC. The present study attempted to identify the effect of piperlongumine on EPCs' angiogenic potential and cerebral ischemic injury in high-fat diet-fed (HFD-fed) mice. Here, we showed that treatment with low-does piperlongumine (0.25 mg/kg/day) for 8 weeks significantly improved EPCs function and reduced the cerebral ischemic injury (both infarct volumes and neurobehavioral outcomes) in HFD-fed mice. In addition, low-dose piperlongumine administration increased intracellular NO level and reduced intracellular O2 - level in EPCs of HFD-fed mice. Moreover, incubation with piperlongumine (1.0 μM, 24 h) reduced thrombospondin-1/2 (TSP-1/2, a potent angiogenesis inhibitor) expression levels in EPCs from HFD-fed mice, increased the therapeutic effect of EPC from HFD-fed mice on cerebral ischemic injury reduction and angiogenesis promotion in HFD-fed mice, and the donor derived EPCs homed to the recipient ischemic brain. In conclusion, low-dose piperlongumine can enhance EPCs' angiogenic potential and protect against cerebral ischemic injury in HFD-fed mice. It is implied that treatment with low-dose piperlongumine might be a potential option to prevent ischemic diseases (including stroke) in patients with hyperlipidemia, and priming with piperlongumine might be a feasible way to improve the efficacy of EPC-based therapy for ischemic diseases.
Keywords: angiogenesis; cerebral ischemic injury; endothelial progenitor cells; hyperlipidemia; piperlongumine.
Copyright © 2021 Dong, Peng, Zhang, Jiang, Yang, He, Tao, Zhang, Chen and Xie.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Low-dose nifedipine rescues impaired endothelial progenitor cell-mediated angiogenesis in diabetic mice.Acta Pharmacol Sin. 2023 Jan;44(1):44-57. doi: 10.1038/s41401-022-00948-w. Epub 2022 Jul 26. Acta Pharmacol Sin. 2023. PMID: 35882957 Free PMC article.
-
AMPK deficiency inhibits fatty acid oxidation in endothelial progenitor cells to aggravate impaired angiogenesis after ischemic stroke in hyperlipidemic mice.Brain Inj. 2024 Aug 23;38(10):835-847. doi: 10.1080/02699052.2024.2349776. Epub 2024 May 8. Brain Inj. 2024. PMID: 38716911
-
Prolonged Fasting Improves Endothelial Progenitor Cell-Mediated Ischemic Angiogenesis in Mice.Cell Physiol Biochem. 2016;40(3-4):693-706. doi: 10.1159/000452581. Epub 2016 Nov 30. Cell Physiol Biochem. 2016. PMID: 27898404
-
Neovascularization and tissue regeneration by endothelial progenitor cells in ischemic stroke.Neurol Sci. 2021 Sep;42(9):3585-3593. doi: 10.1007/s10072-021-05428-3. Epub 2021 Jul 3. Neurol Sci. 2021. PMID: 34216308 Review.
-
Endothelial progenitor cells in ischemic stroke: an exploration from hypothesis to therapy.J Hematol Oncol. 2015 Apr 11;8:33. doi: 10.1186/s13045-015-0130-8. J Hematol Oncol. 2015. PMID: 25888494 Free PMC article. Review.
Cited by
-
Unraveling the potential of endothelial progenitor cells as a treatment following ischemic stroke.Front Neurol. 2022 Sep 8;13:940682. doi: 10.3389/fneur.2022.940682. eCollection 2022. Front Neurol. 2022. PMID: 36158970 Free PMC article. Review.
-
Piperlongumine regulates genes involved in the skin barrier in epidermal keratinocyte HaCaT cells.Anim Cells Syst (Seoul). 2024 Jun 25;28(1):326-339. doi: 10.1080/19768354.2024.2361144. eCollection 2024. Anim Cells Syst (Seoul). 2024. PMID: 38933927 Free PMC article.
References
-
- Baigent C., Keech A., Kearney P. M., Blackwell L., Buck G., Pollicino C., et al. (2005). Efficacy and Safety of Cholesterol-Lowering Treatment: Prospective Meta-Analysis of Data from 90,056 Participants in 14 Randomised Trials of Statins. Lancet 366, 1267–1278. 10.1016/S0140-6736(05)67394-1 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

