Sustained Release of VEGF to Promote Angiogenesis and Osteointegration of Three-Dimensional Printed Biomimetic Titanium Alloy Implants
- PMID: 34869265
- PMCID: PMC8634467
- DOI: 10.3389/fbioe.2021.757767
Sustained Release of VEGF to Promote Angiogenesis and Osteointegration of Three-Dimensional Printed Biomimetic Titanium Alloy Implants
Abstract
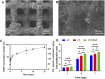
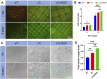
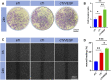
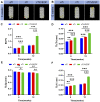
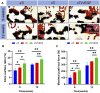
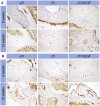
Tumor resection and treatment of trauma-related regional large bone defects have major challenges in the field of orthopedics. Scaffolds that treat bone defects are the focus of bone tissue engineering. 3D printing porous titanium alloy scaffolds, prepared via electron beam melting technology, possess customized structure and strength. The addition of a growth factor coating to the scaffold introduces a specific form of biological activation. Vascular endothelial growth factor (VEGF) is key to angiogenesis and osteogenesis in vivo. We designed a porous titanium alloy scaffold/thermosensitive collagen hydrogel system, equipped with VEGF, to promote local osseointegration and angiogenesis. We also verified the VEGF release via thermosensitive collagen and proliferation and induction of the human umbilical vein endothelial cells (HUVECs) via the composite system in vitro. In vivo, using microscopic computed tomography (Micro-CT), histology, and immunohistochemistry analysis, we confirmed that the composite scaffold aids in angiogenesis-mediated bone regeneration, and promotes significantly more bone integration. We also discovered that the composite scaffold has excellent biocompatibility, provides bioactive VEGF for angiogenesis and osteointegration, and provides an important theoretical basis for the restoration of local blood supply and strengthening of bone integration.
Keywords: 3D-printed porous titanium alloy scaffold; VEGF; angiogenesis; bioactive interface; osseointegration.
Copyright © 2021 Li, Liu, Bai, Li, Shang, Zhu, Zhu, Zhu, Che, Wang, Liu and Huang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Construction and osteogenic effects of 3D-printed porous titanium alloy loaded with VEGF/BMP-2 shell-core microspheres in a sustained-release system.Front Bioeng Biotechnol. 2022 Oct 21;10:1028278. doi: 10.3389/fbioe.2022.1028278. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36338136 Free PMC article.
-
Functionalized chitosan hydrogel promotes osseointegration at the interface of3D printed titanium alloy scaffolds.Int J Biol Macromol. 2024 May;266(Pt 1):131169. doi: 10.1016/j.ijbiomac.2024.131169. Epub 2024 Mar 28. Int J Biol Macromol. 2024. PMID: 38554899
-
3D printing of dual-cell delivery titanium alloy scaffolds for improving osseointegration through enhancing angiogenesis and osteogenesis.BMC Musculoskelet Disord. 2021 Aug 27;22(1):734. doi: 10.1186/s12891-021-04617-7. BMC Musculoskelet Disord. 2021. PMID: 34452607 Free PMC article.
-
Advanced Surface Modification for 3D-Printed Titanium Alloy Implant Interface Functionalization.Front Bioeng Biotechnol. 2022 Mar 1;10:850110. doi: 10.3389/fbioe.2022.850110. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35299643 Free PMC article. Review.
-
Functional hydrogel empowering 3D printing titanium alloys.Mater Today Bio. 2024 Dec 24;30:101422. doi: 10.1016/j.mtbio.2024.101422. eCollection 2025 Feb. Mater Today Bio. 2024. PMID: 39830135 Free PMC article. Review.
Cited by
-
Mitigating challenges and expanding the future of vascular tissue engineering-are we there yet?Front Physiol. 2023 Jan 4;13:1079421. doi: 10.3389/fphys.2022.1079421. eCollection 2022. Front Physiol. 2023. PMID: 36685187 Free PMC article. No abstract available.
-
Enhancing osteogenesis and angiogenesis functions for Ti-24Nb-4Zr-8Sn scaffolds with methacrylated gelatin and deferoxamine.Front Bioeng Biotechnol. 2024 Apr 19;12:1372636. doi: 10.3389/fbioe.2024.1372636. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 38707506 Free PMC article.
-
Low-Intensity Pulsed Ultrasound Promotes Osteogenesis in Porous Titanium Alloys Through miR-1187/BMP4 Pathway.FASEB J. 2025 May 15;39(9):e70583. doi: 10.1096/fj.202403395RR. FASEB J. 2025. PMID: 40317956 Free PMC article.
-
Recent advancement in vascularized tissue-engineered bone based on materials design and modification.Mater Today Bio. 2023 Nov 11;23:100858. doi: 10.1016/j.mtbio.2023.100858. eCollection 2023 Dec. Mater Today Bio. 2023. PMID: 38024843 Free PMC article. Review.
-
Bone Tissue Engineering and Nanotechnology: A Promising Combination for Bone Regeneration.Biology (Basel). 2024 Apr 2;13(4):237. doi: 10.3390/biology13040237. Biology (Basel). 2024. PMID: 38666849 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources

