Evaluation of ligand modified poly (N-Isopropyl acrylamide) hydrogel for etiological diagnosis of corneal infection
- PMID: 34871569
- PMCID: PMC9012892
- DOI: 10.1016/j.exer.2021.108881
Evaluation of ligand modified poly (N-Isopropyl acrylamide) hydrogel for etiological diagnosis of corneal infection
Abstract
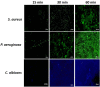
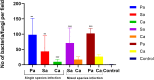
Corneal ulcers, a leading cause of blindness in the developing world are treated inappropriately without prior microbiology assessment because of issues related to availability or cost of accessing these services. In this work we aimed to develop a device for identifying the presence of Gram-positive or Gram-negative bacteria or fungi that can be used by someone without the need for a microbiology laboratory. Working with branched poly (N-isopropyl acrylamide) (PNIPAM) tagged with Vancomycin, Polymyxin B, or Amphotericin B to bind Gram-positive bacteria, Gram-negative bacteria and fungi respectively, grafted onto a single hydrogel we demonstrated specific binding of the organisms. The limit of detection of the microbes by these polymers was between 10 and 4 organisms per high power field (100X) for bacteria and fungi binding polymers respectively. Using ex vivo and animal cornea infection models infected with bacteria, fungi or both we than demonstrated that the triple functionalised hydrogel could pick up all 3 organisms after being in place for 30 min. To confirm the presence of bacteria and fungi we used conventional microbiology techniques and fluorescently labelled ligands or dyes. While we need to develop an easy-to-use either a colorimetric or an imaging system to detect the fluorescent signals, this study presents for the first time a simple to use hydrogel system, which can be applied to infected eyes and specifically binds different classes of infecting agents within a short space of time. Ultimately this diagnostic system will not require trained microbiologists for its use and will be used at the point-of-care.
Keywords: Amphotericin B; Corneal infections; Ex vivo corneal infection model; Fluorescence imaging; Hydrogel; Poly N isopropyl Acrylamide (PNIPAM); Polymyxin B; Vancomycin.
Copyright © 2021 Elsevier Ltd. All rights reserved.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures










References
-
- Barnes R.A. Early diagnosis of fungal infection in immunocompromised patients. J. Antimicrob. Chemother. 2008;61(Suppl. 1):i3–6. - PubMed
-
- Betlem K., Kaur A., Hudson A.D., Crapnell R.D., Hurst G., Singla P., Zubko M., Tedesco S., Banks C.E., Whitehead K., Peeters M. Heat-transfer method: a thermal analysis technique for the real-time monitoring of Staphylococcus aureus growth in buffered solutions and digestate samples. ACS Appl. Bio Mater. 2019;2:3790–3798. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

