Development of mRNA manufacturing for vaccines and therapeutics: mRNA platform requirements and development of a scalable production process to support early phase clinical trials
- PMID: 34871810
- PMCID: PMC8641981
- DOI: 10.1016/j.trsl.2021.11.009
Development of mRNA manufacturing for vaccines and therapeutics: mRNA platform requirements and development of a scalable production process to support early phase clinical trials
Abstract
The remarkable success of SARS CoV-2 mRNA-based vaccines and the ensuing interest in mRNA vaccines and therapeutics have highlighted the need for a scalable clinical-enabling manufacturing process to produce such products, and robust analytical methods to demonstrate safety, potency, and purity. To date, production processes have either not been disclosed or are bench-scale in nature and cannot be readily adapted to clinical and commercial scale production. To address these needs, we have advanced an aqueous-based scalable process that is readily adaptable to GMP-compliant manufacturing, and developed the required analytical methods for product characterization, quality control release, and stability testing. We also have demonstrated the products produced at manufacturing scale under such approaches display good potency and protection in relevant animal models with mRNA products encoding both vaccine immunogens and antibodies. Finally, we discuss continued challenges in raw material identification, sourcing and supply, and the cold chain requirements for mRNA therapeutic and vaccine products. While ultimate solutions have yet to be elucidated, we discuss approaches that can be taken that are aligned with regulatory guidance.
Copyright © 2021. Published by Elsevier Inc.
Figures


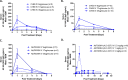
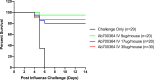
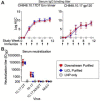
References
-
- Sahin U, Karikó K, Türeci Ö. mRNA-based therapeutics — developing a new class of drugs. Nat Rev Drug Discov. 2014;13:759–780. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

