Measuring Ambulation, Motor, and Behavioral Outcomes with Post-stroke Fluoxetine in Tanzania: The Phase II MAMBO Trial
- PMID: 34872059
- PMCID: PMC8922504
- DOI: 10.4269/ajtmh.21-0653
Measuring Ambulation, Motor, and Behavioral Outcomes with Post-stroke Fluoxetine in Tanzania: The Phase II MAMBO Trial
Abstract
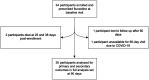
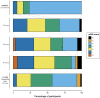
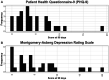
We test the safety of fluoxetine post-ischemic stroke in sub-Saharan Africa. Adults with acute ischemic stroke, seen <14 days since new-onset motor deficits, were enrolled from November 2019 to October 2020 in a single-arm, open-label phase II trial of daily fluoxetine 20 mg for 90 days at Muhimbili National Hospital, Dar es Salaam, Tanzania. The primary outcome was safety with secondary outcomes of medication adherence and tolerability. Thirty-four patients were enrolled (11 were female; mean age 52.2 years, 65% < 60 years old; mean 3.3 days since symptom onset). Participants had hypertension (74%), diabetes (18%), and smoked cigarettes (18%). The median National Institutes of Health Stroke Scale score at enrollment was 10.5. The median Fugl-Meyer Motor Scale score was 28.5 (upper extremity 8, lower extremity 17.5). 32/34 participants (91%) survived to 90 days. There were eight serious and two nonserious adverse events. Deaths occurred due to gastrointestinal illness with low serum sodium (nadir 120 mmol/L) with seizure and gastrointestinal bleed from gastric cancer. The average sodium level at 90 days was 139 mmol/L (range 133-146) and alanine transaminase was 28 U/L (range 10-134). Fluoxetine adherence was 96%. The median modified Rankin Scale score among survivors at 90 days was 2 and Fugl-Meyer Motor Scale score was 66 (upper extremity 40, lower extremity 27). Median 90-day Patient Health Questionnaire-9 and Montgomery-Åsberg scores were 3.5 and 4 (minimal depression). Fluoxetine administration for 90 days poststroke in sub-Saharan Africa was generally safe and well-tolerated, but comorbid illness presentations were fatal in 2/34 cases, even after careful participant selection.
Figures
References
-
- Avendano M Kawachi I Van Lenthe F Boshuizen HC Mackenbach JP Van den Bos GA Fay ME Berkman LF , 2006. Socioeconomic status and stroke incidence in the US elderly: the role of risk factors in the EPESE study. Stroke 37: 1368–1373. - PubMed
-
- Chollet F et al. 2011. Fluoxetine for motor recovery after acute ischaemic stroke (FLAME): a randomised placebo-controlled trial. Lancet Neurol 10: 123–130. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

