RPL35 promotes neuroblastoma progression via the enhanced aerobic glycolysis
- PMID: 34873488
- PMCID: PMC8640819
RPL35 promotes neuroblastoma progression via the enhanced aerobic glycolysis
Erratum in
-
Erratum: RPL35 promotes neuroblastoma progression via the enhanced aerobic glycolysis.Am J Cancer Res. 2022 Dec 15;12(12):5696. eCollection 2022. Am J Cancer Res. 2022. PMID: 36628271 Free PMC article.
Abstract
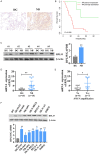
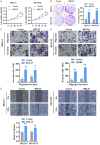
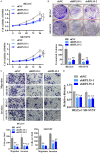
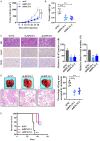
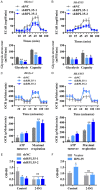
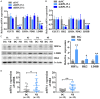
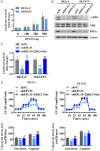
Neuroblastoma (NB) is an rare type of tumor that almost affects children age 5 or younger due to its rapid proliferation ability. The overall survival rate of patients with advanced NB is not satisfactory. Ribosomal proteins (RPs) play a critical role in the development and progress of cancer. However, the contribution of RPL35 in NB has not been proven. In this study, we reveal that RPL35 is upregulated in NB tissues and the upregulation of RPL35 promotes proliferation and migration of NB while RPL35 knockdown significantly restrained the proliferation of NB cells. In terms of mechanism, glycolysis was decreased and the mitochondrial respiration was increased with knockdown of RPL35 in NB cells, indicating that RPL35 function as a positive regulator in aerobic glycolysis. Importantly, our data indicated that RPL35 deficiency decreased HIF1α expression both in mRNA and protein levels. Western blot analysis showed that RPL35 knockdown has a negative regulatory effect on the ERK pathway, and RPL35 modulated aerobic glycolysis in part through its regulation of the RPL35/ERK/HIF1α axis. Overall, RPL35 functions as a positive regulator of aerobic glycolysis, and the RPL35/ERK/HIF1α axis could be a potential therapeutic target for the therapy of NB.
Keywords: ERK; HIF1α; Neuroblastoma; RPL35; aerobic glycolysis.
AJCR Copyright © 2021.
Conflict of interest statement
None.
Figures
Similar articles
-
FUBP1 promotes neuroblastoma proliferation via enhancing glycolysis-a new possible marker of malignancy for neuroblastoma.J Exp Clin Cancer Res. 2019 Sep 11;38(1):400. doi: 10.1186/s13046-019-1414-6. J Exp Clin Cancer Res. 2019. PMID: 31511046 Free PMC article.
-
Klotho negatively regulated aerobic glycolysis in colorectal cancer via ERK/HIF1α axis.Cell Commun Signal. 2018 Jun 8;16(1):26. doi: 10.1186/s12964-018-0241-2. Cell Commun Signal. 2018. PMID: 29884183 Free PMC article.
-
LDHA in Neuroblastoma Is Associated with Poor Outcome and Its Depletion Decreases Neuroblastoma Growth Independent of Aerobic Glycolysis.Clin Cancer Res. 2018 Nov 15;24(22):5772-5783. doi: 10.1158/1078-0432.CCR-17-2578. Epub 2018 Jun 20. Clin Cancer Res. 2018. PMID: 29925504
-
circCUX1 promotes neuroblastoma progression and glycolysis by regulating the miR-338-3p/PHF20 axis.Gen Physiol Biophys. 2021 Jan;40(1):17-29. doi: 10.4149/gpb_2020041. Gen Physiol Biophys. 2021. PMID: 33655888
-
Therapeutic targeting of YY1/MZF1 axis by MZF1-uPEP inhibits aerobic glycolysis and neuroblastoma progression.Theranostics. 2020 Jan 1;10(4):1555-1571. doi: 10.7150/thno.37383. eCollection 2020. Theranostics. 2020. PMID: 32042322 Free PMC article.
Cited by
-
RPL35A Downregulation Suppresses Hepatocellular Carcinoma Cell Proliferation via NCAPG2 Inactivation.Cancer Med. 2025 Jun;14(12):e70985. doi: 10.1002/cam4.70985. Cancer Med. 2025. PMID: 40552444 Free PMC article.
-
Specialized Ribosomes in Health and Disease.Int J Mol Sci. 2023 Mar 28;24(7):6334. doi: 10.3390/ijms24076334. Int J Mol Sci. 2023. PMID: 37047306 Free PMC article. Review.
-
Excavation of gene markers associated with pancreatic ductal adenocarcinoma based on interrelationships of gene expression.IET Syst Biol. 2024 Dec;18(6):261-270. doi: 10.1049/syb2.12090. Epub 2024 Mar 26. IET Syst Biol. 2024. PMID: 38530028 Free PMC article.
-
GADD45GIP1 promotes osteosarcoma progression by modulating RPL35 ubiquitination and alleviating endoplasmic reticulum stress via the PERK/eIF2α pathway.Cancer Cell Int. 2025 Jul 2;25(1):242. doi: 10.1186/s12935-025-03866-z. Cancer Cell Int. 2025. PMID: 40604925 Free PMC article.
-
Discovery, evaluation and mechanism study of WDR5-targeted small molecular inhibitors for neuroblastoma.Acta Pharmacol Sin. 2023 Apr;44(4):877-887. doi: 10.1038/s41401-022-00999-z. Epub 2022 Oct 7. Acta Pharmacol Sin. 2023. PMID: 36207403 Free PMC article.
References
-
- Brodeur GM. Neuroblastoma: biological insights into a clinical enigma. Nat Rev Cancer. 2003;3:203–216. - PubMed
-
- Park JR, Eggert A, Caron H. Neuroblastoma: biology, prognosis, and treatment. Hematol Oncol Clin North Am. 2010;24:65–86. - PubMed
-
- Verissimo CS, Molenaar JJ, Fitzsimons CP, Vreugdenhil E. Neuroblastoma therapy: what is in the pipeline? Endocr Relat Cancer. 2011;18:R213–231. - PubMed
-
- Nakagawara A, Li Y, Izumi H, Muramori K, Inada H, Nishi M. Neuroblastoma. Jpn J Clin Oncol. 2018;48:214–241. - PubMed
-
- Aygun N. Biological and genetic features of neuroblastoma and their clinical importance. Curr Pediatr Rev. 2018;14:73–90. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
