Non-beta blocker enantiomers of propranolol and atenolol inhibit vasculogenesis in infantile hemangioma
- PMID: 34874911
- PMCID: PMC8803322
- DOI: 10.1172/JCI151109
Non-beta blocker enantiomers of propranolol and atenolol inhibit vasculogenesis in infantile hemangioma
Abstract
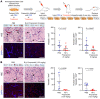
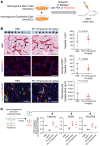
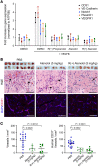
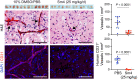
Propranolol and atenolol, current therapies for problematic infantile hemangioma (IH), are composed of R(+) and S(-) enantiomers: the R(+) enantiomer is largely devoid of beta blocker activity. We investigated the effect of R(+) enantiomers of propranolol and atenolol on the formation of IH-like blood vessels from hemangioma stem cells (HemSCs) in a murine xenograft model. Both R(+) enantiomers inhibited HemSC vessel formation in vivo. In vitro, similar to R(+) propranolol, both atenolol and its R(+) enantiomer inhibited HemSC to endothelial cell differentiation. As our previous work implicated the transcription factor sex-determining region Y (SRY) box transcription factor 18 (SOX18) in propranolol-mediated inhibition of HemSC to endothelial differentiation, we tested in parallel a known SOX18 small-molecule inhibitor (Sm4) and show that this compound inhibited HemSC vessel formation in vivo with efficacy similar to that seen with the R(+) enantiomers. We next examined how R(+) propranolol alters SOX18 transcriptional activity. Using a suite of biochemical, biophysical, and quantitative molecular imaging assays, we show that R(+) propranolol directly interfered with SOX18 target gene trans-activation, disrupted SOX18-chromatin binding dynamics, and reduced SOX18 dimer formation. We propose that the R(+) enantiomers of widely used beta blockers could be repurposed to increase the efficiency of current IH treatment and lower adverse associated side effects.
Keywords: Angiogenesis; Drug therapy; Endothelial cells; Transcription; Vascular Biology.
Figures
Comment in
-
A transcription factor is the target of propranolol treatment in infantile hemangioma.J Clin Invest. 2022 Feb 1;132(3):e156863. doi: 10.1172/JCI156863. J Clin Invest. 2022. PMID: 35104803 Free PMC article.
Similar articles
-
R(+) Propranolol decreases lipid accumulation in hemangioma-derived stem cells.bioRxiv [Preprint]. 2024 Jul 2:2024.07.01.601621. doi: 10.1101/2024.07.01.601621. bioRxiv. 2024. Update in: Br J Dermatol. 2025 Mar 18;192(4):757-759. doi: 10.1093/bjd/ljae452. PMID: 39005472 Free PMC article. Updated. Preprint.
-
An endothelial SOX18-mevalonate pathway axis enables repurposing of statins for infantile hemangioma.J Clin Invest. 2025 Feb 25;135(7):e179782. doi: 10.1172/JCI179782. J Clin Invest. 2025. PMID: 39998898 Free PMC article.
-
Propranolol Targets Hemangioma Stem Cells via cAMP and Mitogen-Activated Protein Kinase Regulation.Stem Cells Transl Med. 2016 Jan;5(1):45-55. doi: 10.5966/sctm.2015-0076. Epub 2015 Nov 16. Stem Cells Transl Med. 2016. PMID: 26574555 Free PMC article.
-
β-blocker therapy for infantile hemangioma.Expert Rev Clin Pharmacol. 2020 Aug;13(8):899-915. doi: 10.1080/17512433.2020.1788938. Expert Rev Clin Pharmacol. 2020. PMID: 32662682 Review.
-
Efficacy and safety of oral atenolol for the treatment of infantile haemangioma: A systematic review.Australas J Dermatol. 2019 Aug;60(3):181-185. doi: 10.1111/ajd.12966. Epub 2018 Dec 4. Australas J Dermatol. 2019. PMID: 30515761
Cited by
-
A promising case of preclinical-clinical translation: β-adrenoceptor blockade from the oxygen-induced retinopathy model to retinopathy of prematurity.Front Physiol. 2024 Jun 13;15:1408605. doi: 10.3389/fphys.2024.1408605. eCollection 2024. Front Physiol. 2024. PMID: 38938747 Free PMC article. Review.
-
Global research trends of infantile hemangioma: A bibliometric and visualization analysis from 2000 to 2022.Heliyon. 2023 Oct 21;9(11):e21300. doi: 10.1016/j.heliyon.2023.e21300. eCollection 2023 Nov. Heliyon. 2023. PMID: 37920523 Free PMC article.
-
A transcription factor is the target of propranolol treatment in infantile hemangioma.J Clin Invest. 2022 Feb 1;132(3):e156863. doi: 10.1172/JCI156863. J Clin Invest. 2022. PMID: 35104803 Free PMC article.
-
Infantile hemangioma models: is the needle in a haystack?J Transl Med. 2023 May 6;21(1):308. doi: 10.1186/s12967-023-04144-0. J Transl Med. 2023. PMID: 37149592 Free PMC article. Review.
-
Unveiling the cholesterol-hemangioma axis: a path to new treatments.J Clin Invest. 2025 Apr 1;135(7):e193039. doi: 10.1172/JCI193039. J Clin Invest. 2025. PMID: 40166936 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

