Aqueous proteins help predict the response of patients with neovascular age-related macular degeneration to anti-VEGF therapy
- PMID: 34874918
- PMCID: PMC8759792
- DOI: 10.1172/JCI144469
Aqueous proteins help predict the response of patients with neovascular age-related macular degeneration to anti-VEGF therapy
Abstract
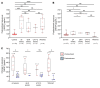
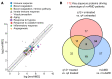
BackgroundTo reduce the treatment burden for patients with neovascular age-related macular degeneration (nvAMD), emerging therapies targeting vascular endothelial growth factor (VEGF) are being designed to extend the interval between treatments, thereby minimizing the number of intraocular injections. However, which patients will benefit from longer-acting agents is not clear.MethodsEyes with nvAMD (n = 122) underwent 3 consecutive monthly injections with currently available anti-VEGF therapies, followed by a treat-and-extend protocol. Patients who remained quiescent 12 weeks from their prior treatment entered a treatment pause and were switched to pro re nata (PRN) treatment (based on vision, clinical exam, and/or imaging studies). Proteomic analysis was performed on aqueous fluid to identify proteins that correlate with patients' response to treatment.ResultsAt the end of 1 year, 38 of 122 eyes (31%) entered a treatment pause (≥30 weeks). Conversely, 21 of 122 eyes (17%) failed extension and required monthly treatment at the end of year 1. Proteomic analysis of aqueous fluid identified proteins that correlated with patients' response to treatment, including proteins previously implicated in AMD pathogenesis. Interestingly, apolipoprotein-B100 (ApoB100), a principal component of drusen implicated in the progression of nonneovascular AMD, was increased in treated patients who required less frequent injections. ApoB100 expression was higher in AMD eyes compared with controls but was lower in eyes that develop choroidal neovascularization (CNV), consistent with a protective role. Accordingly, mice overexpressing ApoB100 were partially protected from laser-induced CNV.FundingThis work was supported by the National Eye Institute, National Institutes of Health grants R01EY029750, R01EY025705, and R01 EY27961; the Research to Prevent Blindness, Inc.; the Alcon Research Institute; and Johns Hopkins University through the Robert Bond Welch and Branna and Irving Sisenwein professorships in ophthalmology.ConclusionAqueous biomarkers could help identify patients with nvAMD who may not require or benefit from long-term treatment with anti-VEGF therapy.
Keywords: Clinical practice; Complement; Expression profiling; Ophthalmology.
Conflict of interest statement
Figures






References
-
- Nguyen DH, et al. Current therapeutic approaches in neovascular age-related macular degeneration. Discov Med. 2013;15(85):343–348. - PubMed
-
- Senger DR, et al. A highly conserved vascular permeability factor secreted by a variety of human and rodent tumor cell lines. Cancer Res. 1986;46(11):5629–5632. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

