Catalase deficiency facilitates the shuttling of free fatty acid to brown adipose tissue through lipolysis mediated by ROS during sustained fasting
- PMID: 34876210
- PMCID: PMC8650429
- DOI: 10.1186/s13578-021-00710-5
Catalase deficiency facilitates the shuttling of free fatty acid to brown adipose tissue through lipolysis mediated by ROS during sustained fasting
Abstract
Background: Fatty acids (FA) derived from adipose tissue and liver serve as the main fuel in thermogenesis of brown adipose tissue (BAT). Catalase, a peroxisomal enzyme, plays an important role in maintaining intracellular redox homeostasis by decomposing hydrogen peroxide to either water or oxygen that oxidize and provide fuel for cellular metabolism. Although the antioxidant enzymatic activity of catalase is well known, its role in the metabolism and maintenance of energy homeostasis has not yet been revealed. The present study investigated the role of catalase in lipid metabolism and thermogenesis during nutrient deprivation in catalase-knockout (KO) mice.
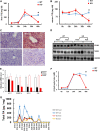
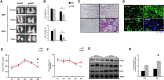
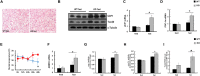
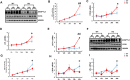
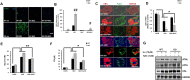
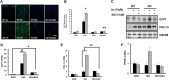
Results: We found that hepatic triglyceride accumulation in KO mice decreased during sustained fasting due to lipolysis through reactive oxygen species (ROS) generation in adipocytes. Furthermore, the free FA released from lipolysis were shuttled to BAT through the activation of CD36 and catabolized by lipoprotein lipase in KO mice during sustained fasting. Although the exact mechanism for the activation of the FA receptor enzyme, CD36 in BAT is still unclear, we found that ROS generation in adipocytes mediated the shuttling of FA to BAT.
Conclusions: Taken together, our findings uncover the novel role of catalase in lipid metabolism and thermogenesis in BAT, which may be useful in understanding metabolic dysfunction.
Keywords: Catalase; Lipolysis; Reactive Oxygen species (ROS); Sustained fasting; Thermogenesis.
© 2021. The Author(s).
Conflict of interest statement
The authors have no competing interest to declare.
Figures
References
-
- Heeren J, Scheja L. Brown adipose tissue and lipid metabolism. Curr Opin Lipidol. 2018;29(3):180–185. - PubMed
-
- Finn PF, Dice JF. Proteolytic and lipolytic responses to starvation. Nutrition. 2006;22(7–8):830–844. - PubMed
-
- Lee JN, Dutta RK, Kim SG, Lim JY, Kim SJ, Choe SK, Yoo KW, Song SR, Park DS, So HS. Fenofibrate, a peroxisome proliferator-activated receptor α ligand, prevents abnormal liver function induced by a fasting-refeeding process. Biochem Biophys Res Commun. 2013;442(1–2):22–27. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

