Current and Future Tools for Diagnosis of Kaposi's Sarcoma
- PMID: 34885035
- PMCID: PMC8657166
- DOI: 10.3390/cancers13235927
Current and Future Tools for Diagnosis of Kaposi's Sarcoma
Abstract
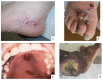
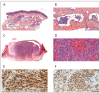
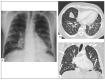
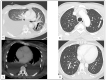
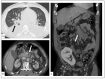
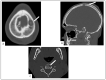
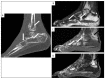
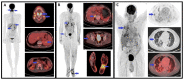
Kaposi's sarcoma (KS) is a rare, atypical malignancy associated with immunosuppression and can be qualified as an opportunistic tumor, which responds to immune modulation or restoration. Four different epidemiological forms have been individualized (AIDS-related, iatrogenic, endemic or classic KS). Although clinical examination is sufficient to diagnose cutaneous lesions of KS, additional explorations are necessary in order to detect lesions involving other organs. New histological markers have been developed in recent years concerning the detection of HHV-8 latent or lytic proteins in the lesions, helping to confirm the diagnosis when it is clinically doubtful. More recently, the evaluation of the local immune response has also been shown to provide some guidance in choosing the appropriate therapeutic option when necessary. We also review the indication and the results of conventional radiological imaging and of non-invasive imaging tools such as 18F-fluoro-deoxy-glucose positron emission tomography, thermography and laser Doppler imaging for the diagnosis of KS and for the follow-up of therapeutic response in patients requiring systemic treatment.
Keywords: FDG-PET; Kaposi’s sarcoma; diagnostic; non-invasive imaging tools; staging.
Conflict of interest statement
All authors have no conflict of interest to declare.
Figures

References
-
- Grabar S., Abraham B., Mahamat A., Del Giudice P., Rosenthal E., Costagliola D. Differential Impact of Combination Antiretroviral Therapy in Preventing Kaposi’s Sarcoma with and without Visceral Involvement. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2006;24:3408–3414. doi: 10.1200/JCO.2005.05.4072. - DOI - PubMed
-
- Hleyhel M., Belot A., Bouvier A.M., Tattevin P., Pacanowski J., Genet P., De Castro N., Berger J.-L., Dupont C., Lavolé A., et al. Risk of AIDS-Defining Cancers among HIV-1-Infected Patients in France between 1992 and 2009: Results from the FHDH-ANRS CO4 Cohort. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2013;57:1638–1647. doi: 10.1093/cid/cit497. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

