Antimicrobial Properties of Chitosan and Chitosan Derivatives in the Treatment of Enteric Infections
- PMID: 34885715
- PMCID: PMC8659174
- DOI: 10.3390/molecules26237136
Antimicrobial Properties of Chitosan and Chitosan Derivatives in the Treatment of Enteric Infections
Abstract
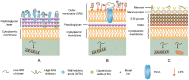
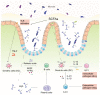
Antibiotics played an important role in controlling the development of enteric infection. However, the emergence of antibiotic resistance and gut dysbiosis led to a growing interest in the use of natural antimicrobial agents as alternatives for therapy and disinfection. Chitosan is a nontoxic natural antimicrobial polymer and is approved by GRAS (Generally Recognized as Safe by the United States Food and Drug Administration). Chitosan and chitosan derivatives can kill microbes by neutralizing negative charges on the microbial surface. Besides, chemical modifications give chitosan derivatives better water solubility and antimicrobial property. This review gives an overview of the preparation of chitosan, its derivatives, and the conjugates with other polymers and nanoparticles with better antimicrobial properties, explains the direct and indirect mechanisms of action of chitosan, and summarizes current treatment for enteric infections as well as the role of chitosan and chitosan derivatives in the antimicrobial agents in enteric infections. Finally, we suggested future directions for further research to improve the treatment of enteric infections and to develop more useful chitosan derivatives and conjugates.
Keywords: antibacterial; antifungal; antimicrobial; antimicrobial mechanisms of chitosan; chitosan; colonization resistance; drug delivery; enteric infection; gut microbiota.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Antimicrobial Chitosan and Chitosan Derivatives: A Review of the Structure-Activity Relationship.Biomacromolecules. 2017 Nov 13;18(11):3846-3868. doi: 10.1021/acs.biomac.7b01058. Epub 2017 Oct 3. Biomacromolecules. 2017. PMID: 28933147 Review.
-
Current state on the development of nanoparticles for use against bacterial gastrointestinal pathogens. Focus on chitosan nanoparticles loaded with phenolic compounds.Carbohydr Polym. 2015 Oct 5;130:429-39. doi: 10.1016/j.carbpol.2015.05.030. Epub 2015 May 21. Carbohydr Polym. 2015. PMID: 26076644 Review.
-
Chitosan Derivatives Active against Multidrug-Resistant Bacteria and Pathogenic Fungi: In Vivo Evaluation as Topical Antimicrobials.Mol Pharm. 2016 Oct 3;13(10):3578-3589. doi: 10.1021/acs.molpharmaceut.6b00764. Epub 2016 Sep 21. Mol Pharm. 2016. PMID: 27589087
-
Chitosan derivatives with antimicrobial, antitumour and antioxidant activities--a review.Curr Pharm Des. 2011;17(32):3596-607. doi: 10.2174/138161211798194468. Curr Pharm Des. 2011. PMID: 22074429 Review.
-
Antimicrobial activity of chitosan derivatives containing N-quaternized moieties in its backbone: a review.Int J Mol Sci. 2014 Nov 13;15(11):20800-32. doi: 10.3390/ijms151120800. Int J Mol Sci. 2014. PMID: 25402643 Free PMC article. Review.
Cited by
-
Self-Assembled Amphiphilic Chitosan Nanomicelles: Synthesis, Characterization and Antibacterial Activity.Biomolecules. 2023 Oct 30;13(11):1595. doi: 10.3390/biom13111595. Biomolecules. 2023. PMID: 38002276 Free PMC article.
-
Temperature Induced Gelation and Antimicrobial Properties of Pluronic F127 Based Systems.Polymers (Basel). 2023 Jan 10;15(2):355. doi: 10.3390/polym15020355. Polymers (Basel). 2023. PMID: 36679236 Free PMC article.
-
Hydrogels in Cutaneous Wound Healing: Insights into Characterization, Properties, Formulation and Therapeutic Potential.Gels. 2024 Mar 8;10(3):188. doi: 10.3390/gels10030188. Gels. 2024. PMID: 38534606 Free PMC article. Review.
-
Studies of the Tarragon Essential Oil Effects on the Characteristics of Doped Hydroxyapatite/Chitosan Biocomposites.Polymers (Basel). 2023 Apr 16;15(8):1908. doi: 10.3390/polym15081908. Polymers (Basel). 2023. PMID: 37112055 Free PMC article.
-
Iron oxide nanoparticles coated with bioactive materials: a viable theragnostic strategy to improve osteosarcoma treatment.Discov Nano. 2025 Jan 30;20(1):18. doi: 10.1186/s11671-024-04163-w. Discov Nano. 2025. PMID: 39883285 Free PMC article. Review.
References
-
- Saberpour M., Bakhshi B., Najar-Peerayeh S. Evaluation of the Antimicrobial and Antibiofilm Effect of Chitosan Nanoparticles as Carrier for Supernatant of Mesenchymal Stem Cells on Multidrug-Resistant Vibrio cholerae. Infect. Drug Resist. 2020;13:2251–2260. doi: 10.2147/IDR.S244990. - DOI - PMC - PubMed
-
- Bennett J.E., Dolin R., Blaser M.J. Syndromes of Enteric Infection. In: LaRocque R.C., Harris J., editors. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. 9th ed. Saunders Elsevier; Philadelphia, PA, USA: 2019. pp. 1330–1339.
-
- Marder E.P., Cieslak P.R., Cronquist A.B., Dunn J., Lathrop S., Rabatsky-Ehr T., Ryan P., Smith K., Tobin-D’Angelo M., Vugia D.J., et al. Incidence and Trends of Infections with Pathogens Transmitted Commonly Through Food and the Effect of Increasing Use of Culture-Independent Diagnostic Tests on Surveillance-Foodborne Diseases Active Surveillance Network, 10 U.S. Sites, 2013–2016. MMWR. Morb. Mortal. Wkly. Rep. 2017;66:397–403. doi: 10.15585/mmwr.mm6615a1. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

