The Anthelmintic Quassinoids Ailanthone and Bruceine a Induce Infertility in the Model Organism Caenorhabditis elegans by an Apoptosis-like Mechanism Induced in Gonadal and Spermathecal Tissues
- PMID: 34885936
- PMCID: PMC8659173
- DOI: 10.3390/molecules26237354
The Anthelmintic Quassinoids Ailanthone and Bruceine a Induce Infertility in the Model Organism Caenorhabditis elegans by an Apoptosis-like Mechanism Induced in Gonadal and Spermathecal Tissues
Abstract
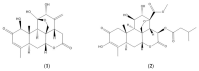
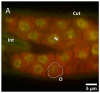
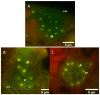
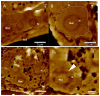
In continuation of the search for new anthelmintic natural products, the study at hand investigated the nematicidal effects of the two naturally occurring quassinoids ailanthone and bruceine A against the reproductive system of the model nematode Caenorhabditis elegans to pinpoint their anthelmintic mode of action by the application of various microscopic techniques. Differential Interference Contrast (DIC) and the epifluorescence microscopy experiments used in the presented study indicated the genotoxic effects of the tested quassinoids (c ailanthone = 50 µM, c bruceine A = 100 µM) against the nuclei of the investigated gonadal and spermathecal tissues, leaving other morphological key features such as enterocytes or body wall muscle cells unimpaired. In order to gain nanoscopic insight into the morphology of the gonads as well as the considerably smaller spermathecae of C. elegans, an innovative protocol of polyethylene glycol embedding, ultra-sectioning, acridine orange staining, tissue identification by epifluorescence, and subsequent AFM-based ultrastructural data acquisition was applied. This sequence allowed the facile and fast assessment of the impact of quassinoid treatment not only on the gonadal but also on the considerably smaller spermathecal tissues of C. elegans. These first-time ultrastructural investigations on C. elegans gonads and spermathecae by AFM led to the identification of specific quassinoid-induced alterations to the nuclei of the reproductive tissues (e.g., highly condensed chromatin, impaired nuclear membrane morphology, as well as altered nucleolus morphology), altogether implying an apoptosis-like effect of ailanthone and bruceine A on the reproductive tissues of C. elegans.
Keywords: Caenorhabditis elegans; ailanthone; anthelmintic natural products; atomic force microscopy; bruceine A; polyethylene glycol embedding; quassinoid; ultramicrotomy; ultrastructural morphology.
Conflict of interest statement
The authors declare no conflict of interest.
Figures








Similar articles
-
Extract and the quassinoid ailanthone from Ailanthus altissima inhibit nematode reproduction by damaging germ cells and rachis in the model organism Caenorhabditis elegans.Fitoterapia. 2020 Oct;146:104651. doi: 10.1016/j.fitote.2020.104651. Epub 2020 Jun 3. Fitoterapia. 2020. PMID: 32504655
-
Caenorhabditis elegans revisited by atomic force microscopy - Ultra-structural changes of the cuticle, but not in the intestine after treatment with Combretum mucronatum extract.J Struct Biol. 2019 Nov 1;208(2):174-181. doi: 10.1016/j.jsb.2019.08.013. Epub 2019 Aug 30. J Struct Biol. 2019. PMID: 31476367
-
In vivo anthelmintic activity of bruceine A and bruceine D from Brucea javanica against Dactylogyrus intermedius (Monogenea) in goldfish (Carassius auratus).Vet Parasitol. 2011 Apr 19;177(1-2):127-33. doi: 10.1016/j.vetpar.2010.11.040. Epub 2010 Dec 3. Vet Parasitol. 2011. PMID: 21196080
-
Anthelmintic drugs and nematicides: studies in Caenorhabditis elegans.WormBook. 2014 Dec 16:1-29. doi: 10.1895/wormbook.1.143.2. WormBook. 2014. PMID: 25517625 Free PMC article. Review.
-
Caenorhabditis elegans in anthelmintic research - Old model, new perspectives.Int J Parasitol Drugs Drug Resist. 2020 Dec;14:237-248. doi: 10.1016/j.ijpddr.2020.09.005. Epub 2020 Oct 2. Int J Parasitol Drugs Drug Resist. 2020. PMID: 33249235 Free PMC article. Review.
Cited by
-
Natural Products for Drug Discovery in the 21st Century: Innovations for Novel Therapeutics.Molecules. 2023 Apr 25;28(9):3690. doi: 10.3390/molecules28093690. Molecules. 2023. PMID: 37175100 Free PMC article.
-
Enterococcus faecalis SI-FC-01 enhances the stress resistance and healthspan of C. elegans via AKT signaling pathway.Sci Rep. 2025 Apr 25;15(1):14454. doi: 10.1038/s41598-025-98440-y. Sci Rep. 2025. PMID: 40281021 Free PMC article.
References
-
- World Health Organization Fact Sheet—Soil-Transmitted Helminth Infections. [(accessed on 14 November 2021)]. Available online: https://www.who.int/en/news-room/fact-sheets/detail/soil-transmitted-hel....
-
- Agyare C., Spiegler V., Sarkodie H., Asase A., Liebau E., Hensel A. An ethnopharmacological survey and in vitro confirmation of the ethnopharmacological use of medicinal plants as anthelmintic remedies in the Ashanti region, in the central part of Ghana. Pt AJ. Ethnopharmacol. 2014;158:255–263. doi: 10.1016/j.jep.2014.10.029. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

