JMM Profile: Carbapenems: a broad-spectrum antibiotic
- PMID: 34889726
- PMCID: PMC8744278
- DOI: 10.1099/jmm.0.001462
JMM Profile: Carbapenems: a broad-spectrum antibiotic
Abstract
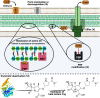
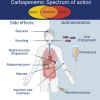
Carbapenems are potent members of the β-lactam family that inhibit bacterial cell-wall biosynthesis inhibitors . They are highly effective against Gram-negative and Gram-positive drug-resistant infections . As such, carbapenems are typically reserved as an antibiotic of last resort. The WHO lists meropenem as an essential medicine. Nausea and vomiting are reported in ≤20% of carbapenem recipients, with 1.5% suffering seizures. Enzymatic hydrolysis of the β-lactam ring is the main driver of clinical resistance. These enzymes can be classified as Class A, B and D. Classes A and D are serine β-lactamases, whereas Class B rely on metal-mediated hydrolysis, typically through zinc.
Keywords: antimicrobial resistance; last resort; β-lactams.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures
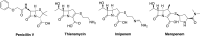
Similar articles
-
Comparative review of the carbapenems.Drugs. 2007;67(7):1027-52. doi: 10.2165/00003495-200767070-00006. Drugs. 2007. PMID: 17488146 Review.
-
Cefiderocol: A Siderophore Cephalosporin with Activity Against Carbapenem-Resistant and Multidrug-Resistant Gram-Negative Bacilli.Drugs. 2019 Feb;79(3):271-289. doi: 10.1007/s40265-019-1055-2. Drugs. 2019. PMID: 30712199 Review.
-
QM/MM Simulations Reveal the Determinants of Carbapenemase Activity in Class A β-Lactamases.ACS Infect Dis. 2022 Aug 12;8(8):1521-1532. doi: 10.1021/acsinfecdis.2c00152. Epub 2022 Jul 25. ACS Infect Dis. 2022. PMID: 35877936 Free PMC article.
-
A plethora of carbapenem resistance in Acinetobacter baumannii: no end to a long insidious genetic journey.J Chemother. 2021 May;33(3):137-155. doi: 10.1080/1120009X.2020.1847421. Epub 2020 Nov 27. J Chemother. 2021. PMID: 33243098 Review.
-
Sulfamoyl Heteroarylcarboxylic Acids as Promising Metallo-β-Lactamase Inhibitors for Controlling Bacterial Carbapenem Resistance.mBio. 2020 Mar 17;11(2):e03144-19. doi: 10.1128/mBio.03144-19. mBio. 2020. PMID: 32184250 Free PMC article.
Cited by
-
A Real-World Study on the Clinical Characteristics, Outcomes, and Relationship between Antibiotic Exposure and Clostridioides difficile Infection.Antibiotics (Basel). 2024 Feb 1;13(2):144. doi: 10.3390/antibiotics13020144. Antibiotics (Basel). 2024. PMID: 38391530 Free PMC article.
-
Insights on the performance of phenotypic tests versus genotypic tests for the detection of carbapenemase-producing Gram-negative bacilli in resource-limited settings.BMC Microbiol. 2022 Oct 14;22(1):248. doi: 10.1186/s12866-022-02660-5. BMC Microbiol. 2022. PMID: 36229768 Free PMC article.
-
Agents Targeting the Bacterial Cell Wall as Tools to Combat Gram-Positive Pathogens.Molecules. 2024 Aug 27;29(17):4065. doi: 10.3390/molecules29174065. Molecules. 2024. PMID: 39274911 Free PMC article. Review.
-
Enhancing effect of natural adjuvant, panduratin A, on antibacterial activity of colistin against multidrug-resistant Acinetobacter baumannii.Sci Rep. 2024 Apr 29;14(1):9863. doi: 10.1038/s41598-024-60627-0. Sci Rep. 2024. PMID: 38684853 Free PMC article.
-
Difference analysis and characteristics of incompatibility group plasmid replicons in gram-negative bacteria with different antimicrobial phenotypes in Henan, China.BMC Microbiol. 2024 Feb 19;24(1):64. doi: 10.1186/s12866-024-03212-9. BMC Microbiol. 2024. PMID: 38373913 Free PMC article.
References
-
- Trebosc V, Schellhorn B, Schill J, Lucchini V, Bühler J, et al. In vitro activity of rifabutin against 293 contemporary carbapenem-resistant Acinetobacter baumannii clinical isolates and characterization of rifabutin mode of action and resistance mechanisms. J Antimicrob Chemother. 2020;75:3552–3562. doi: 10.1093/jac/dkaa370. - DOI - PMC - PubMed
-
- Yocum RR, Rasmussen JR, Strominger JL. The mechanism of action of penicillin. Penicillin acylates the active site of Bacillus stearothermophilus D-alanine carboxypeptidase. J Biol Chem. 1980;255:3977–3986. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

