Concordant Androgen-Regulated Expression of Divergent Rhox5 Promoters in Sertoli Cells
- PMID: 34902009
- PMCID: PMC8667857
- DOI: 10.1210/endocr/bqab237
Concordant Androgen-Regulated Expression of Divergent Rhox5 Promoters in Sertoli Cells
Abstract
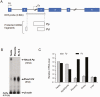
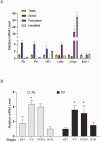
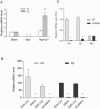
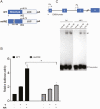
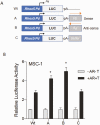
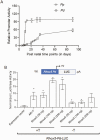
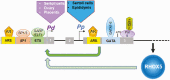
Concordant transcriptional regulation can generate multiple gene products that collaborate to achieve a common goal. Here we report a case of concordant transcriptional regulation that instead drives a single protein to be produced in the same cell type from divergent promoters. This gene product-the RHOX5 homeobox transcription factor-is translated from 2 different mRNAs with different 5' untranslated regions (UTRs) transcribed from alternative promoters. Despite the fact that these 2 promoters-the proximal promoter (Pp) and the distal promoter (Pd)-exhibit different patterns of tissue-specific activity, share no obvious sequence identity, and depend on distinct transcription factors for expression, they exhibit a remarkably similar expression pattern in the testes. In particular, both depend on androgen signaling for expression in the testes, where they are specifically expressed in Sertoli cells and have a similar stage-specific expression pattern during the seminiferous epithelial cycle. We report evidence for 3 mechanisms that collaborate to drive concordant Pp/Pd expression. First, both promoters have an intrinsic ability to respond to androgen receptor and androgen. Second, the Pp acts as an enhancer to promote androgen-dependent transcription from the Pd. Third, Pd transcription is positively autoregulated by the RHOX5 protein, which is first produced developmentally from the Pp. Together, our data support a model in which the Rhox5 homeobox gene evolved multiple mechanisms to activate both of its promoters in Sertoli cells to produce Rhox5 in an androgen-dependent manner during different phases of spermatogenesis.
Keywords: ARE; Rhox5; Sertoli; androgen receptor; gene regulation; transcription factors.
© The Author(s) 2021. Published by Oxford University Press on behalf of the Endocrine Society. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures


Similar articles
-
Androgen-induced Rhox homeobox genes modulate the expression of AR-regulated genes.Mol Endocrinol. 2010 Jan;24(1):60-75. doi: 10.1210/me.2009-0303. Epub 2009 Nov 9. Mol Endocrinol. 2010. PMID: 19901196 Free PMC article.
-
DNA demethylation-dependent AR recruitment and GATA factors drive Rhox5 homeobox gene transcription in the epididymis.Mol Endocrinol. 2012 Apr;26(4):538-49. doi: 10.1210/me.2011-1059. Epub 2012 Feb 9. Mol Endocrinol. 2012. PMID: 22322598 Free PMC article.
-
Defective survival of proliferating Sertoli cells and androgen receptor function in a mouse model of the ATR-X syndrome.Hum Mol Genet. 2011 Jun 1;20(11):2213-24. doi: 10.1093/hmg/ddr109. Epub 2011 Mar 22. Hum Mol Genet. 2011. PMID: 21427128
-
Epigenetic regulation and downstream targets of the Rhox5 homeobox gene.Int J Androl. 2008 Sep;31(5):462-70. doi: 10.1111/j.1365-2605.2008.00904.x. Epub 2008 Jul 11. Int J Androl. 2008. PMID: 18637153 Review.
-
Transcriptional regulation of cell adhesion at the blood-testis barrier and spermatogenesis in the testis.Adv Exp Med Biol. 2012;763:281-94. doi: 10.1007/978-1-4614-4711-5_14. Adv Exp Med Biol. 2012. PMID: 23397630 Free PMC article. Review.
Cited by
-
Updates in Sertoli Cell-Mediated Signaling During Spermatogenesis and Advances in Restoring Sertoli Cell Function.Front Endocrinol (Lausanne). 2022 May 4;13:897196. doi: 10.3389/fendo.2022.897196. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35600584 Free PMC article. Review.
-
What Does Androgen Receptor Signaling Pathway in Sertoli Cells During Normal Spermatogenesis Tell Us?Front Endocrinol (Lausanne). 2022 Feb 24;13:838858. doi: 10.3389/fendo.2022.838858. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35282467 Free PMC article. Review.
References
-
- Davuluri RV, Suzuki Y, Sugano S, Plass C, Huang TH. The functional consequences of alternative promoter use in mammalian genomes. Trends Genet. 2008;24(4):167-177. - PubMed
-
- Demircioğlu D, Cukuroglu E, Kindermans M, et al. . A pan-cancer transcriptome analysis reveals pervasive regulation through alternative promoters. Cell. 2019;178(6):1465-1477.e17. - PubMed
-
- Carninci P, Sandelin A, Lenhard B, et al. . Genome-wide analysis of mammalian promoter architecture and evolution. Nat Genet. 2006;38(6):626-635. - PubMed
-
- Sandelin A, Carninci P, Lenhard B, Ponjavic J, Hayashizaki Y, Hume DA. Mammalian RNA polymerase II core promoters: insights from genome-wide studies. Nat Rev Genet. 2007;8(6):424-436. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

