Cardiovascular Safety Assessment in Cancer Drug Development
- PMID: 34913360
- PMCID: PMC9075223
- DOI: 10.1161/JAHA.121.024033
Cardiovascular Safety Assessment in Cancer Drug Development
Abstract
The development of cardiovascular toxicity attributable to anticancer drugs is a pivotal event that is associated with cardiovascular morbidity as well as with worse cancer-specific and overall outcomes. Although broad consensus exists regarding the importance of cardiovascular safety assessment in cancer drug development, real-world data suggest that cardiovascular events are significantly underestimated in oncology trials. This drug safety discrepancy has profound implications on drug development decisions, risk-benefit evaluation, formulation of surveillance and prevention protocols, and survivorship. In this article, we review the contemporary cardiovascular safety evaluation of new pharmaceuticals in hematology and oncology, spanning from in vitro pharmacodynamic testing to randomized clinical trials. We argue that cardiovascular safety assessment of anticancer drugs should be reformed and propose practical strategies, including development and validation of preclinical assays, expansion of oncology trial eligibility, incorporation of cardiovascular end points in early-phase studies, and design of longitudinal multi-institutional cardiotoxicity registries.
Keywords: antineoplastic agents; cardiotoxicity; cardiovascular safety; consensus; randomized controlled trials.
Figures
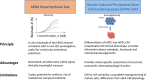

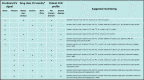

Similar articles
-
Strategies to Prevent Cardiotoxicity.Curr Treat Options Oncol. 2020 Apr 8;21(4):32. doi: 10.1007/s11864-020-0722-6. Curr Treat Options Oncol. 2020. PMID: 32270293 Review.
-
Essentials of cardio-oncology.Clin Med (Lond). 2023 Jan;23(1):52-55. doi: 10.7861/clinmed.2022-0588. Clin Med (Lond). 2023. PMID: 36697021 Free PMC article.
-
Cardio-Oncology: An Update on Cardiotoxicity of Cancer-Related Treatment.Circ Res. 2016 Mar 18;118(6):1008-20. doi: 10.1161/CIRCRESAHA.115.303633. Circ Res. 2016. PMID: 26987914 Review.
-
Drug-induced cardiac toxicity and adverse drug reactions, a narrative review.Therapie. 2024 Mar-Apr;79(2):161-172. doi: 10.1016/j.therap.2023.10.008. Epub 2023 Oct 31. Therapie. 2024. PMID: 37957054 Review.
-
Cancer drug related cardiotoxicity during breast cancer treatment.Expert Opin Drug Saf. 2016 Aug;15(8):1063-74. doi: 10.1080/14740338.2016.1182493. Epub 2016 Jul 6. Expert Opin Drug Saf. 2016. PMID: 27120499 Review.
Cited by
-
Cardiovascular Safety in Oncology Clinical Trials: JACC: CardioOncology Primer.JACC CardioOncol. 2025 Feb;7(2):83-95. doi: 10.1016/j.jaccao.2024.09.014. Epub 2024 Dec 10. JACC CardioOncol. 2025. PMID: 39967209 Free PMC article. Review.
-
Advancing Cardio-Oncology in Asia.Korean Circ J. 2023 Feb;53(2):69-91. doi: 10.4070/kcj.2022.0255. Korean Circ J. 2023. PMID: 36792558 Free PMC article. Review.
-
Biotechnological advances and applications of human pluripotent stem cell-derived heart models.Front Bioeng Biotechnol. 2023 Jul 25;11:1214431. doi: 10.3389/fbioe.2023.1214431. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 37560538 Free PMC article. Review.
-
Advancing the care of individuals with cancer through innovation & technology: Proceedings from the cardiology oncology innovation summit 2020 and 2021.Am Heart J Plus. 2023 Dec 25;38:100354. doi: 10.1016/j.ahjo.2023.100354. eCollection 2024 Feb. Am Heart J Plus. 2023. PMID: 38510746 Free PMC article.
-
KMU-191 Induces Apoptosis in Human Clear Cell Renal Cell Carcinoma Caki Cells Through Modulation of Bcl-xL, Mcl-1 (L), c-FLIP (L), and p53 Proteins.J Cancer. 2023 Jul 16;14(12):2224-2235. doi: 10.7150/jca.85650. eCollection 2023. J Cancer. 2023. PMID: 37576393 Free PMC article.
References
-
- Ball S, Ghosh RK, Wongsaengsak S, Bandyopadhyay D, Ghosh GC, Aronow WS, Fonarow GC, Lenihan DJ, Bhatt DL. Cardiovascular toxicities of immune checkpoint inhibitors: JACC review topic of the week. J Am Coll Cardiol. 2019;74:1714–1727. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

