The toxin mimic FS48 from the salivary gland of Xenopsylla cheopis functions as a Kv1.3 channel-blocking immunomodulator of T cell activation
- PMID: 34919963
- PMCID: PMC8732088
- DOI: 10.1016/j.jbc.2021.101497
The toxin mimic FS48 from the salivary gland of Xenopsylla cheopis functions as a Kv1.3 channel-blocking immunomodulator of T cell activation
Abstract
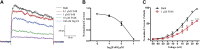
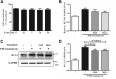
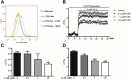
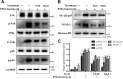
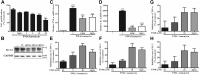
The Kv1.3 channel has been widely demonstrated to play crucial roles in the activation and proliferation of T cells, which suggests that selective blockers could serve as potential therapeutics for autoimmune diseases mediated by T cells. We previously described that the toxin mimic FS48 from salivary gland of Xenopsylla cheopis downregulates the secretion of proinflammatory factors by Raw 264.7 cells by blocking the Kv1.3 channel and the subsequent inactivation of the proinflammatory MAPK/NF-κB pathways. However, the effects of FS48 on human T cells and autoimmune diseases are unclear. Here, we described its immunomodulatory effects on human T cells derived from suppression of Kv1.3 channel. Kv1.3 currents in Jurkat T cells were recorded by whole-cell patch-clamp, and Ca2+ influx, cell proliferation, and TNF-α and IL-2 secretion were measured using Fluo-4, CCK-8, and ELISA assays, respectively. The in vivo immunosuppressive activity of FS48 was evaluated with a rat DTH model. We found that FS48 reduced Kv1.3 currents in Jurkat T cells in a concentration-dependent manner with an IC50 value of about 1.42 μM. FS48 also significantly suppressed Kv1.3 protein expression, Ca2+ influx, MAPK/NF-κB/NFATc1 pathway activation, and TNF-α and IL-2 production in activated Jurkat T cells. Finally, we show that FS48 relieved the DTH response in rats. We therefore conclude that FS48 can block the Kv1.3 channel and inhibit human T cell activation, which most likely contributes to its immunomodulatory actions and highlights the great potential of this evolutionary-guided peptide as a drug template in future studies.
Keywords: FS48; Jurkat T cells; Xenopsylla cheopis; autoimmune disease; immunomodulator; potassium channel.
Copyright © 2021 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of interest The authors declare that they have no conflicts of interest with the contents of this article.
Figures
Similar articles
-
Anti-inflammatory effects of FS48, the first potassium channel inhibitor from the salivary glands of the flea Xenopsylla cheopis.J Biol Chem. 2021 Jan-Jun;296:100670. doi: 10.1016/j.jbc.2021.100670. Epub 2021 Apr 15. J Biol Chem. 2021. PMID: 33864815 Free PMC article.
-
Acacetin blocks kv1.3 channels and inhibits human T cell activation.Cell Physiol Biochem. 2014;34(4):1359-72. doi: 10.1159/000366343. Epub 2014 Oct 2. Cell Physiol Biochem. 2014. PMID: 25301362
-
The Scorpion Toxin Analogue BmKTX-D33H as a Potential Kv1.3 Channel-Selective Immunomodulator for Autoimmune Diseases.Toxins (Basel). 2016 Apr 19;8(4):115. doi: 10.3390/toxins8040115. Toxins (Basel). 2016. PMID: 27104568 Free PMC article.
-
Toxins Targeting the Kv1.3 Channel: Potential Immunomodulators for Autoimmune Diseases.Toxins (Basel). 2015 May 19;7(5):1749-64. doi: 10.3390/toxins7051749. Toxins (Basel). 2015. PMID: 25996605 Free PMC article. Review.
-
Peptide blockers of Kv1.3 channels in T cells as therapeutics for autoimmune disease.Curr Opin Chem Biol. 2017 Jun;38:97-107. doi: 10.1016/j.cbpa.2017.02.015. Epub 2017 Apr 14. Curr Opin Chem Biol. 2017. PMID: 28412597 Review.
Cited by
-
An insight into the draft genome of the Oriental rat flea, Xenopsylla cheopis, together with its Wolbachia endosymbiont.BMC Genomics. 2025 Jul 1;26(1):621. doi: 10.1186/s12864-025-11759-8. BMC Genomics. 2025. PMID: 40597597 Free PMC article.
-
Metabolism and immune responses of striped hamsters to ectoparasite challenges: insights from transcriptomic analysis.Front Immunol. 2024 Dec 12;15:1516382. doi: 10.3389/fimmu.2024.1516382. eCollection 2024. Front Immunol. 2024. PMID: 39723213 Free PMC article.
-
Revisiting the sialome of the cat flea Ctenocephalides felis.PLoS One. 2023 Jan 17;18(1):e0279070. doi: 10.1371/journal.pone.0279070. eCollection 2023. PLoS One. 2023. PMID: 36649293 Free PMC article.
-
Transcriptome analysis of Kunming mice responses to the bite of Xenopsylla cheopis.Parasit Vectors. 2024 Jun 7;17(1):250. doi: 10.1186/s13071-024-06331-4. Parasit Vectors. 2024. PMID: 38849919 Free PMC article.
References
-
- Feske S. Calcium signalling in lymphocyte activation and disease. Nat. Rev. Immunol. 2007;7:690–702. - PubMed
-
- Desai R., Peretz A., Idelson H., Lazarovici P., Attali B. Ca2+-activated K+ channels in human leukemic Jurkat T cells. Molecular cloning, biochemical and functional characterization. J. Biol. Chem. 2000;275:39954–39963. - PubMed
-
- Fu X.X., Du L.L., Zhao N., Dong Q., Liao Y.H., Du Y.M. 18beta-Glycyrrhetinic acid potently inhibits Kv1.3 potassium channels and T cell activation in human Jurkat T cells. J. Ethnopharmacol. 2013;148:647–654. - PubMed
-
- Zhao N., Dong Q., Fu X.X., Du L.L., Cheng X., Du Y.M., Liao Y.H. Acacetin blocks kv1.3 channels and inhibits human T cell activation. Cell. Physiol. Biochem. 2014;34:1359–1372. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

