Adaptive Dosing of Nivolumab + Ipilimumab Immunotherapy Based Upon Early, Interim Radiographic Assessment in Advanced Melanoma (The ADAPT-IT Study)
- PMID: 34928709
- PMCID: PMC8966970
- DOI: 10.1200/JCO.21.01570
Adaptive Dosing of Nivolumab + Ipilimumab Immunotherapy Based Upon Early, Interim Radiographic Assessment in Advanced Melanoma (The ADAPT-IT Study)
Abstract
Purpose: Nivolumab + ipilimumab (nivo + ipi) is highly efficacious but has high toxicity. Standard treatment in advanced melanoma is four doses of nivo + ipi followed by nivo alone. Whether four doses of nivo + ipi are needed is unclear.
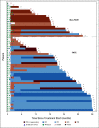
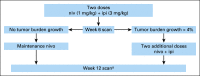
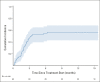
Methods: The Adaptively Dosed ImmunoTherapy Trial (ADAPT-IT) study (NCT03122522) is a multicenter, single-arm phase II clinical trial. Patients received two doses of nivo (1 mg/kg) + ipi (3 mg/kg) followed by a computed tomography scan at week 6. Patients without new lesions or index lesion tumor growth of > 4% had protocol-defined early favorable antitumor effect (FATE) and ceased nivo + ipi, transitioning to nivo monotherapy. Patients without FATE at week 6 received the standard third and fourth doses of nivo + ipi followed by nivo monotherapy. The primary end point was response rate by RECIST 1.1 at week 12. Secondary end points included additional efficacy assessments and safety.
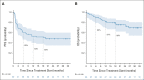
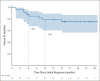
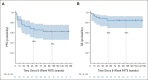
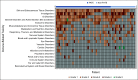
Results: Sixty patients were enrolled; 41 patients (68%) had FATE at week 6 and met criteria for stopping nivo + ipi. Best overall response rates by RECIST at week 12 or any time afterward were 48% (95% CI, 35 to 62) and 58% (95% CI, 45 to 71), respectively. With a median follow-up of 25 months, the estimated 18-month progression-free survival and overall survival are 52% and 80%, respectively. Fifty seven percent of patients had grade 3-5 treatment-related toxicity.
Conclusion: The efficacy and toxicity of standard four dose nivo + ipi induction therapy in melanoma is likely driven by the first two doses. An interim computed tomography scan after two doses guided cessation of combination dosing and identified almost all responders. Longer follow-up and further study are needed to fully understand the implications of a shortened induction course of nivo + ipi.
Conflict of interest statement
Figures
References
-
- Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Five-year survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 2019;381:1535–1546. - PubMed
-
- Zimmer L, Livingstone E, Hassel JC, et al. Adjuvant nivolumab plus ipilimumab or nivolumab monotherapy versus placebo in patients with resected stage IV melanoma with no evidence of disease (IMMUNED): A randomised, double-blind, placebo-controlled, phase 2 trial. Lancet. 2020;395:1558–1568. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

