In Vitro and In Vivo Activity of (Trifluoromethyl)pyridines as Anti- Chlamydia trachomatis Agents
- PMID: 34935346
- PMCID: PMC9516413
- DOI: 10.1021/acsinfecdis.1c00553
In Vitro and In Vivo Activity of (Trifluoromethyl)pyridines as Anti- Chlamydia trachomatis Agents
Abstract
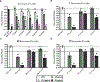
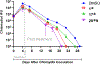
Chlamydia trachomatis is the leading pathogen in sexually transmitted bacterial infections across the globe. The development of a selective treatment against this pathogen could be an attractive therapeutic option that will reduce the overuse of broad-spectrum antibiotics. Previously, we reported some sulfonylpyridine-based compounds that showed selectivity against C. trachomatis. Here, we describe a set of related compounds that display enhanced anti-chlamydial potency when compared to our early leads. We found that the active molecules are bactericidal and have no impact on Staphylococcus aureus or Escherichia coli strains. Importantly, the molecules were not toxic to mammalian cells. Furthermore, a combination of molecule 20 (the most active molecule) and azithromycin at subinhibitory concentrations acted synergistically to inhibit chlamydial growth. Molecule 20 also eradicated Chlamydia in a 3D infection model and accelerated the recovery of Chlamydia-infected mice. This work presents compounds that could be further developed to be used alone or in combination with existing treatment regimens against chlamydial infections.
Keywords: 3D culture; Chlamydia trachomatis; sexually transmitted diseases.
Conflict of interest statement
The authors declare no competing financial interest.
Figures






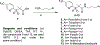
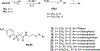
References
-
- Unemo M; Bradshaw CS; Hocking JS; de Vries HJ; Francis SC; Mabey D; Marrazzo JM; Sonder GJ; Schwebke JR; Hoornenborg E Sexually Transmitted Infections: Challenges Ahead. Lancet Infect. Dis 2017, 17, e235–e279. - PubMed
-
- Dallabetta G; Wi T; Nielsen G; Holmes K; Sparling P; Stamm W Prevention and Control of STD and HIV Infection in Developing Countries. Sexually Transmitted Diseases, 4th ed.; McGraw-Hill: New York, NY, 2008.
-
- World Health Organization. Report on Global Sexually Transmitted Infection Surveillance 2018. 2018, https://www.who.int/reproductivehealth/publications/stis-surveillance-20... (accessed Dec 07, 2021).
-
- Yoneyama H; Katsumata R Antibiotic Resistance in Bacteria and Its Future for Novel Antibiotic Development. Biosci., Biotechnol., Biochem 2006, 70, 1060–1075. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

