Immune and Genome Engineering as the Future of Transplantable Tissue
- PMID: 34936741
- PMCID: PMC9009215
- DOI: 10.1056/NEJMra1913421
Immune and Genome Engineering as the Future of Transplantable Tissue
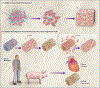
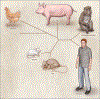
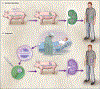
Figures

References
-
- Viebahn CS, Benseler V, Holz LE, et al. Invading macrophages play a major role in the liver progenitor cell response to chronic liver injury. J Hepatol 2010;53: 500–7. - PubMed
-
- Doyle AM, Lechler RI, Turka LA. Organ transplantation: halfway through the first century. J Am Soc Nephrol 2004;15: 2965–71. - PubMed
-
- Kissane NA, Itani KMF. A decade of ventral incisional hernia repairs with biologic acellular dermal matrix: what have we learned? Plast Reconstr Surg 2012;130: Suppl 2:194S–202S. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
