Enhancing Endocannabinoid Control of Stress with Cannabidiol
- PMID: 34945148
- PMCID: PMC8704602
- DOI: 10.3390/jcm10245852
Enhancing Endocannabinoid Control of Stress with Cannabidiol
Abstract
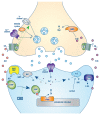
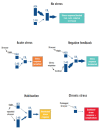
The stress response is a well-defined physiological function activated frequently by life events. However, sometimes the stress response can be inappropriate, excessive, or prolonged; in which case, it can hinder rather than help in coping with the stressor, impair normal functioning, and increase the risk of somatic and mental health disorders. There is a need for a more effective and safe pharmacological treatment that can dampen maladaptive stress responses. The endocannabinoid system is one of the main regulators of the stress response. A basal endocannabinoid tone inhibits the stress response, modulation of this tone permits/curtails an active stress response, and chronic deficiency in the endocannabinoid tone is associated with the pathological complications of chronic stress. Cannabidiol is a safe exogenous cannabinoid enhancer of the endocannabinoid system that could be a useful treatment for stress. There have been seven double-blind placebo controlled clinical trials of CBD for stress on a combined total of 232 participants and one partially controlled study on 120 participants. All showed that CBD was effective in significantly reducing the stress response and was non-inferior to pharmaceutical comparators, when included. The clinical trial results are supported by the established mechanisms of action of CBD (including increased N-arachidonylethanolamine levels) and extensive real-world and preclinical evidence of the effectiveness of CBD for treating stress.
Keywords: AEA; CBD; N-arachidonylethanolamine; cannabidiol; endocannabinoid; hypothalamus–pituitary–adrenal axis; stress.
Conflict of interest statement
The authors declare no conflict of interest. Medlab Clinical Ltd. (Sydney, Australia) funded the publication costs and had no role in the design of the review; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources

