Recent Advances in the Oxone-Mediated Synthesis of Heterocyclic Compounds
- PMID: 34946605
- PMCID: PMC8705989
- DOI: 10.3390/molecules26247523
Recent Advances in the Oxone-Mediated Synthesis of Heterocyclic Compounds
Abstract
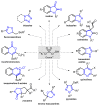
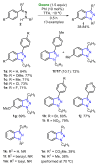
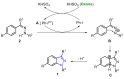
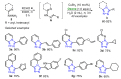
Oxone is a commercially available oxidant, composed of a mixture of three inorganic species, being the potassium peroxymonosulfate (KHSO5) the reactive one. Over the past few decades, this cheap and environmentally friendly oxidant has become a powerful tool in organic synthesis, being extensively employed to mediate the construction of a plethora of important compounds. This review summarizes the recent advances in the Oxone-mediated synthesis of N-, O- and chalcogen-containing heterocyclic compounds, through a wide diversity of reactions, starting from several kinds of substrate, highlighting the main synthetic differences, advantages, the scope and limitations.
Keywords: Oxone; green chemistry; heterocycles; oxidant.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


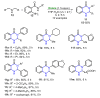
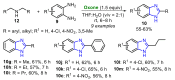
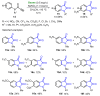
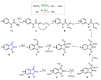
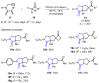
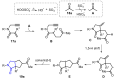

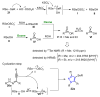
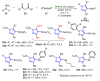








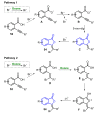
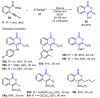
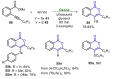
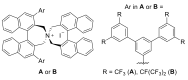






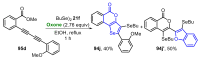
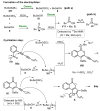
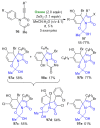
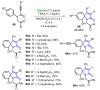
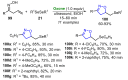


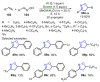
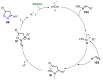
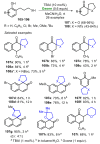
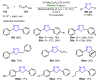
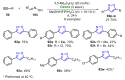
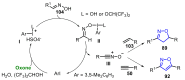
References
-
- Anastas P.T., Warner J.C. Green Chemistry: Theory and Practice. Oxford University Press; New York, NY, USA: 1998.
-
- Lenardão E.J., Freitag R.A., Dabdoub J.M., Batista A.C.F., Silveira C.C. Green Chemistry: The 12 Principles of Green Chemistry and It Insertion in the Teach and Research Activities. Quim. Nova. 2003;26:123–129. doi: 10.1590/S0100-40422003000100020. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

