Pan-Caspase Inhibitor zVAD Induces Necroptotic and Autophagic Cell Death in TLR3/4-Stimulated Macrophages
- PMID: 34949739
- PMCID: PMC9001149
- DOI: 10.14348/molcells.2021.0193
Pan-Caspase Inhibitor zVAD Induces Necroptotic and Autophagic Cell Death in TLR3/4-Stimulated Macrophages
Abstract
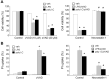
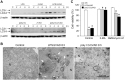
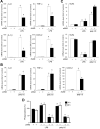
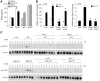
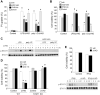
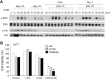
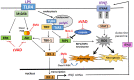
In addition to inducing apoptosis, caspase inhibition contributes to necroptosis and/or autophagy depending on the cell type and cellular context. In macrophages, necroptosis can be induced by co-treatment with Toll-like receptor (TLR) ligands (lipopolysaccharide [LPS] for TLR4 and polyinosinic-polycytidylic acid [poly I:C] for TLR3) and a cell-permeable pan-caspase inhibitor zVAD. Here, we elucidated the signaling pathways and molecular mechanisms of cell death. We showed that LPS/zVAD- and poly I:C/zVAD-induced cell death in bone marrow-derived macrophages (BMDMs) was inhibited by receptor-interacting protein kinase 1 (RIP1) inhibitor necrostatin-1 and autophagy inhibitor 3-methyladenine. Electron microscopic images displayed autophagosome/autolysosomes, and immunoblotting data revealed increased LC3II expression. Although zVAD did not affect LPS- or poly I:C-induced activation of IKK, JNK, and p38, it enhanced IRF3 and STAT1 activation as well as type I interferon (IFN) expression. In addition, zVAD inhibited ERK and Akt phosphorylation induced by LPS and poly I:C. Of note, zVAD-induced enhancement of the IRF3/IFN/STAT1 axis was abolished by necrostatin-1, while zVAD-induced inhibition of ERK and Akt was not. Our data further support the involvement of autocrine IFNs action in reactive oxygen species (ROS)-dependent necroptosis, LPS/zVAD-elicited ROS production was inhibited by necrostatin-1, neutralizing antibody of IFN receptor (IFNR) and JAK inhibitor AZD1480. Accordingly, both cell death and ROS production induced by TLR ligands plus zVAD were abrogated in STAT1 knockout macrophages. We conclude that enhanced TRIF-RIP1-dependent autocrine action of IFNβ, rather than inhibition of ERK or Akt, is involved in TLRs/zVAD-induced autophagic and necroptotic cell death via the JAK/STAT1/ROS pathway.
Keywords: JAK/STAT1; autophagy; interferon; macrophage; necrosis; zVAD.
Conflict of interest statement
The authors have no potential conflicts of interest to disclose.
Figures



References
-
- Bonapace L., Bornhauser B.C., Schmitz M., Cario G., Ziegler U., Niggli F.K., Schäfer B.W., Schrappe M., Stanulla M., Bourquin J.P. Induction of autophagy-dependent necroptosis is required for childhood acute lymphoblastic leukemia cells to overcome glucocorticoid resistance. J. Clin. Invest. 2010;120:1310–1323. doi: 10.1172/JCI39987. - DOI - PMC - PubMed
-
- Chen C.Y., Hung Y.F., Tsai C.Y., Shih Y.C., Chou T.F., Lai M.Z., Wang T.F., Hsueh Y.P. Transcriptomic analysis and C-terminal epitope tagging reveal differential processing and signaling of endogenous TLR3 and TLR7. Front. Immunol. 2021;12:686060. doi: 10.3389/fimmu.2021.686060. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

