Fatty Acid Metabolism and Derived-Mediators Distinctive of PPAR-α Activation in Obese Subjects Post Bariatric Surgery
- PMID: 34959892
- PMCID: PMC8705357
- DOI: 10.3390/nu13124340
Fatty Acid Metabolism and Derived-Mediators Distinctive of PPAR-α Activation in Obese Subjects Post Bariatric Surgery
Abstract
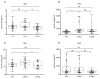
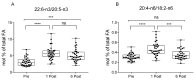
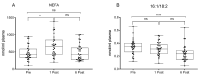
Bariatric surger (BS) is characterized by lipid metabolic changes as a response to the massive release of non-esterified fatty acids (NEFA) from adipose depots. The study aimed at evaluating changes in polyunsaturated fatty acids (PUFA) metabolism and biosynthesis of the lipid mediators N-acylethanolamines (NAE), as indices of nuclear peroxisome proliferator-activated receptor (PPAR)-α activation. The observational study was performed on 35 subjects (27 female, 8 male) with obesity, undergoing bariatric surgery. We assessed plasma FA and NAE profiles by LC-MS/MS, clinical parameters and anthropometric measures before and 1 and 6 months after bariatric surgery. One month after bariatric surgery, as body weight and clinical parameters improved significantly, we found higher plasma levels of N-oleoylethanolamine, arachidonic and a 22:6-n3/20:5-n3 ratio as evidence of PPAR-α activation. These changes corresponded to higher circulating levels of NEFA and a steep reduction of the fat mass. After 6 months 22:6-n3/20:5-n3 remained elevated and fat mass was further reduced. Our data suggest that the massive release of NEFA from adipose tissue at 1-Post, possibly by inducing PPAR-α, may enhance FA metabolism contributing to fat depot reduction and improved metabolic parameters in the early stage. However, PUFA metabolic changes favor n6 PUFA biosynthesis, requiring a nutritional strategy aimed at reducing the n6/n3 PUFA ratio.
Keywords: N-oleoylethanolamine (OEA); bariatric surgery; non-esterified fatty acid (NEFA); obesity; peroxisome proliferator-activated receptor (PPAR)-α.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

