A Fermented Milk Product Containing B. lactis CNCM I-2494 Improves the Tolerance of a Plant-Based Diet in Patients with Disorders of Gut-Brain Interactions
- PMID: 34960094
- PMCID: PMC8709116
- DOI: 10.3390/nu13124542
A Fermented Milk Product Containing B. lactis CNCM I-2494 Improves the Tolerance of a Plant-Based Diet in Patients with Disorders of Gut-Brain Interactions
Abstract
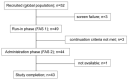
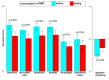
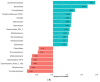
Healthy, plant-based diets, rich in fermentable residues, may induce gas-related symptoms. The aim of this exploratory study was to assess the effects of a fermented milk product, containing probiotics, on the tolerance of a healthy diet in patients with disorders of gut-brain interactions (DGBI), complaining of excessive flatulence. In an open design, a 3-day healthy, mostly plant-based diet was administered to patients with DGBI (52 included, 43 completed) before and at the end of 28 days of consumption of a fermented milk product (FMP) containing Bifidobacterium animalis subsp. lactis CNCM I-2494 and lactic acid bacteria. As compared to a habitual diet, the flatulogenic diet increased the perception of digestive symptoms (flatulence score 7.1 ± 1.6 vs. 5.8 ± 1.9; p < 0.05) and the daily number of anal gas evacuations (22.4 ± 12.5 vs. 16.5 ± 10.2; p < 0.0001). FMP consumption reduced the flatulence sensation score (by -1.6 ± 2.2; p < 0.05) and the daily number of anal gas evacuations (by -5.3 ± 8.2; p < 0.0001). FMP consumption did not significantly alter the overall gut microbiota composition, but some changes in the microbiota correlated with the observed clinical improvement. The consumption of a product containing B. lactis CNCM I-2494 improved the tolerance of a healthy diet in patients with DGBI, and this effect may be mediated, in part, by the metabolic activity of the microbiota.
Keywords: B. lactis DN-173010; digestive symptoms; disorders of gut–brain interactions; fermentable carbohydrates; flatulence; microbiota; probiotics.
Conflict of interest statement
B.L.N., J.T., M.D., J.M.F., A.C. and Q.D. are Danone Nutricia Research employees. F.A. has received unrestricted research grants from Danone Nutricia Research. Other authors have nothing to disclose.
Figures


Similar articles
-
A Fermented Milk Product with B. Lactis CNCM I-2494 and Lactic Acid Bacteria Improves Gastrointestinal Comfort in Response to a Challenge Diet Rich in Fermentable Residues in Healthy Subjects.Nutrients. 2020 Jan 25;12(2):320. doi: 10.3390/nu12020320. Nutrients. 2020. PMID: 31991794 Free PMC article. Clinical Trial.
-
Human gut metatranscriptome changes induced by a fermented milk product are associated with improved tolerance to a flatulogenic diet.Comput Struct Biotechnol J. 2022 Apr 5;20:1632-1641. doi: 10.1016/j.csbj.2022.04.001. eCollection 2022. Comput Struct Biotechnol J. 2022. PMID: 35465165 Free PMC article.
-
Consumption of a Fermented Milk Product Containing Bifidobacterium lactis CNCM I-2494 in Women Complaining of Minor Digestive Symptoms: Rapid Response Which Is Independent of Dietary Fibre Intake or Physical Activity.Nutrients. 2019 Jan 4;11(1):92. doi: 10.3390/nu11010092. Nutrients. 2019. PMID: 30621211 Free PMC article.
-
THE EFFECT OF PROBIOTIC FERMENTED MILK THAT INCLUDES BIFIDOBACTERIUM LACTIS CNCM I-2494 ON THE REDUCTION OF GASTROINTESTINAL DISCOMFORT AND SYMPTOMS IN ADULTS: A NARRATIVE REVIEW.Nutr Hosp. 2015 Aug 1;32(2):501-9. doi: 10.3305/nh.2015.32.2.9232. Nutr Hosp. 2015. PMID: 26268077 Review.
-
Impact of Fermented Milk On Gut Microbiota And Human Health: A Comprehensive Review.Curr Microbiol. 2025 Jan 31;82(3):107. doi: 10.1007/s00284-025-04061-z. Curr Microbiol. 2025. PMID: 39888432 Review.
References
-
- Guyonnet D., Schlumberger A., Mhamdi L., Jakob S., Chassany O. Fermented milk containing bifidobacterium lactis dn-173 010 improves gastrointestinal well-being and digestive symptoms in women reporting minor digestive symptoms: A randomised, double-blind, parallel, controlled study. Br. J. Nutr. 2009;102:1654–1662. doi: 10.1017/S0007114509990882. - DOI - PubMed
-
- Guyonnet D., Woodcock A., Stefani B., Trevisan C., Hall C. Fermented milk containing bifidobacterium lactis dn-173 010 improved self-reported digestive comfort amongst a general population of adults. A randomized, open-label, controlled, pilot study. J. Dig. Dis. 2009;10:61–70. doi: 10.1111/j.1751-2980.2008.00366.x. - DOI - PubMed
-
- Marteau P., Guyonnet D., Lafaye de Micheaux P., Gelu S. A randomized, double-blind, controlled study and pooled analysis of two identical trials of fermented milk containing probiotic bifidobacterium lactis cncm i-2494 in healthy women reporting minor digestive symptoms. Neurogastroenterol. Motil. 2013;25:331-e252. doi: 10.1111/nmo.12078. - DOI - PubMed
-
- Marteau P., Le Neve B., Quinquis L., Pichon C., Whorwell P.J., Guyonnet D. Consumption of a fermented milk product containing bifidobacterium lactis cncm i-2494 in women complaining of minor digestive symptoms: Rapid response which is independent of dietary fibre intake or physical activity. Nutrients. 2019;11:92. doi: 10.3390/nu11010092. - DOI - PMC - PubMed
-
- Agrawal A., Houghton L.A., Morris J., Reilly B., Guyonnet D., Goupil Feuillerat N., Schlumberger A., Jakob S., Whorwell P.J. Clinical trial: The effects of a fermented milk product containing bifidobacterium lactis dn-173 010 on abdominal distension and gastrointestinal transit in irritable bowel syndrome with constipation. Aliment. Pharmacol. Ther. 2009;29:104–114. doi: 10.1111/j.1365-2036.2008.03853.x. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

