Surgical Treatment of Low-Lying Rectal Cancer: Updates
- PMID: 34961303
- PMCID: PMC8717072
- DOI: 10.3393/ac.2021.00927.0132
Surgical Treatment of Low-Lying Rectal Cancer: Updates
Abstract
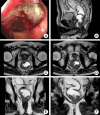
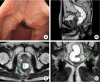
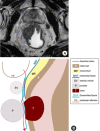
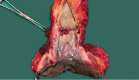
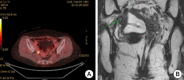
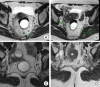
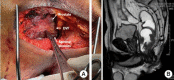
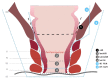
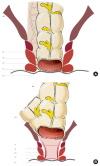
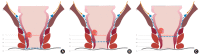
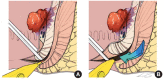
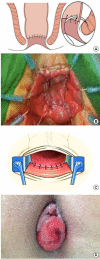
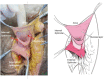
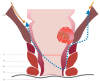
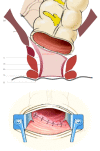
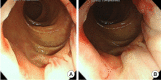
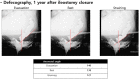
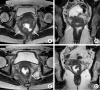
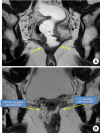
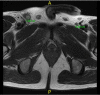
Despite innovative advancements, distally located rectal cancer remains a critical disease of challenging management. The crucial location of the tumor predisposes it to a circumferential resection margin (CRM) that tends to involve the anal sphincter complex and surrounding organs, with a high incidence of delayed anastomotic complications and the risk of the pelvic sidewall or rarely inguinal lymph node metastases. In this regard, colorectal surgeons should be aware of other issues beyond total mesorectal excision (TME) performance. For decades, the concept of extralevator abdominoperineal resection to avoid compromised CRM has been introduced. However, the complexity of deep pelvic dissection with poor visualization in low-lying rectal cancer has led to transanal TME. In contrast, neoadjuvant chemoradiotherapy (NCRT) has allowed for the execution of more sphincter-saving procedures without oncologic compromise. Significant tumor regression after NCRT and complete pathologic response also permit applying the watch-and-wait protocol in some cases, now with more solid evidence. This review article will introduce the current surgical treatment options, their indication and technical details, and recent oncologic and functional outcomes. Lastly, the novel characteristics of distal rectal cancer, such as pelvic sidewall and inguinal lymph node metastases, will be discussed along with its tailored and individualized treatment approach.
Keywords: Lymph node excision; Proctectomy; Rectal neoplasms; Surgical procedures.
Conflict of interest statement
No potential conflict of interest relevant to this article was reported.
Figures


References
-
- Church JM, Raudkivi PJ, Hill GL. The surgical anatomy of the rectum: a review with particular relevance to the hazards of rectal mobilisation. Int J Colorectal Dis. 1987;2:158–66. - PubMed
-
- Bogach J, Tsai S, Zbuk K, Wong R, Grubac V, Coates A, et al. Quality of preoperative pelvic computed tomography (CT) and magnetic resonance imaging (MRI) for rectal cancer in a region in Ontario: a retrospective population-based study. J Surg Oncol. 2018;117:1038–42. - PubMed
-
- Zhou Y, Shao W, Lu W. Diagnostic value of endorectal ultrasonography for rectal carcinoma: a meta-analysis. J Cancer Res Ther. 2014;10 Suppl:319–22. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

