Comprehensive serologic profile and specificity of maternal and neonatal cord blood SARS-CoV-2 antibodies
- PMID: 34961853
- PMCID: PMC8697419
- DOI: 10.1016/j.xagr.2021.100046
Comprehensive serologic profile and specificity of maternal and neonatal cord blood SARS-CoV-2 antibodies
Abstract
Background: Initial studies on COVID-19 in pregnancy have demonstrated a range of neutralizing activity, but little has been published on the full profile of SARS CoV-2 related antibodies in maternal and cordblood.
Objective: This study aimed to describe the profile and specificity of maternal and neonatal cord blood antibody profiles in response to SARS-CoV-2 virus exposure.
Study design: This was a prospective cohort study of delivering patients at Thomas Jefferson University Hospital from April 2020 to February 2021. The primary objective was to describe unique maternal and fetal antibody epitope titers and specificity in patients with COVID-19 history. Serologic profile was assessed with a multiplex platform. Antigens used were hemagglutinin trimer influenza A (Hong Kong H3); spike trimers for SARS-CoV-2, SARS-CoV-1, Middle East respiratory syndrome coronavirus, and betacoronaviruses HKU-1 and OC43; and spike N-terminal domain, spike receptor-binding domain, and nucleocapsid protein (full length) for SARS-CoV-2.
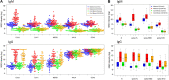
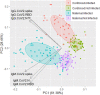
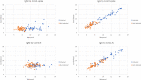
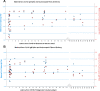
Results: Here, 112 maternal samples and 101 maternal and cord blood pairs were analyzed. Of note, 37 patients had a known history of COVID-19 (positive polymerase chain reaction test) during pregnancy. Of 36 patients, 16 (44%) were diagnosed with COVID-19 within 7 days of delivery. Moreover, 15 of the remaining 76 patients (20%) without a known diagnosis had positive maternal serology. For those with a history of COVID-19, we identified robust immunoglobulin G response in maternal blood to CoV-2 nucleocapsid, spike (full length), and spike (receptor-binding domain) antigens with more modest responses to the spike (N-terminal domain) antigen. In contrast, the maternal blood immunoglobulin M response seemed more specific to spike (full length) epitopes than nucleocapsid, spike (receptor-binding domain), or spike (N-terminal domain) epitopes. There were significantly higher maternal and cord blood immunoglobulin G responses not only to CoV-2 spike (127.1-fold; standard deviation, 2.0; P<.00001) but also to CoV-1 spike (21.1-fold higher; standard deviation, 1.8; P<.00001) and Middle East respiratory syndrome spike (6.9-fold higher; standard deviation, 2.5; P<.00001). In contrast, maternal immunoglobulin M responses were more specific to CoV-2 spike (15.8-fold; standard deviation, 2.1; P<.00001) but less specific to CoV-1 (2.5-fold higher; standard deviation, 0.71; P<.00001) and no significant difference for Middle East respiratory syndrome. Maternal and cord blood immunoglobulin G antibodies were highly correlated for both spike and nucleocapsid (R2=0.96 and 0.94, respectively).
Conclusion: Placental transfer was efficient, with robust nucleocapsid and spike responses. Both nucleocapsid and spike antibody responses should be studied for a better understanding of COVID-19 immunity. Immunoglobulin G antibodies were cross-reactive with related CoV-1 and Middle East respiratory syndrome spike epitopes, whereas immunoglobulin M antibodies, which cannot cross the placenta to provide neonatal passive immunity, were more SARS-CoV-2 specific. Neonatal cord blood may have significantly different fine specificity than maternal blood, despite the high efficiency of immunoglobulin G transfer.
Keywords: COVID-19; immunity; passive immunity; pregnancy; serology.
© 2021 The Authors.
Figures
Update of
-
Comprehensive Serological Profile and Specificity of Maternal and Neonatal Cord Blood SARS CoV-2 Antibodies.medRxiv [Preprint]. 2021 Dec 7:2021.12.06.21267328. doi: 10.1101/2021.12.06.21267328. medRxiv. 2021. Update in: AJOG Glob Rep. 2022 Feb;2(1):100046. doi: 10.1016/j.xagr.2021.100046. PMID: 34909795 Free PMC article. Updated. Preprint.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

